Research Article
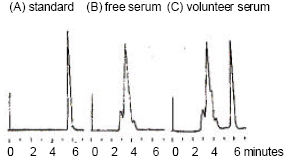
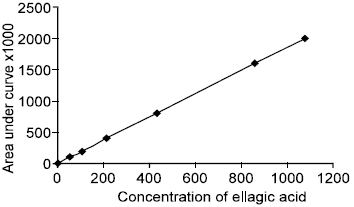
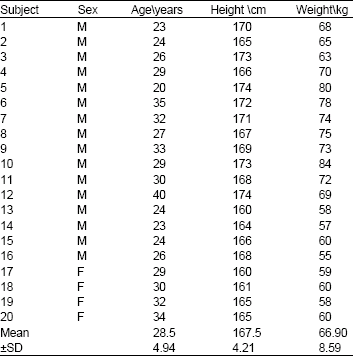
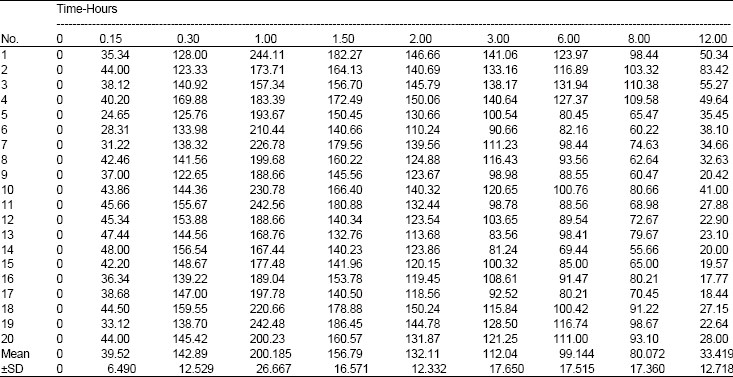
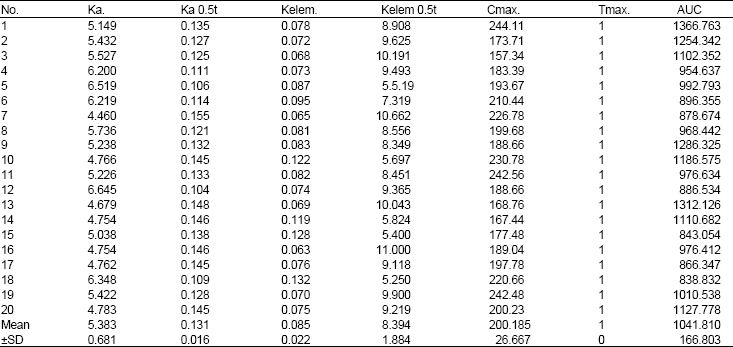
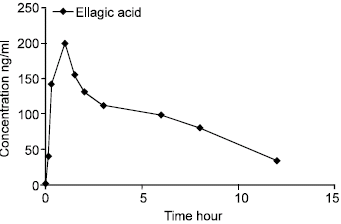
Bioavailability of Ellagic Acid After Single Dose Administration Using HPLC
Department of Allied Medical Sciences, Zarqa University College, Al Balq`Applied University, Al Salt, Jordan
Waleed M. Al-Momani
Department of Allied Medical Sciences, Zarqa University College, Al Balq`Applied University, Al Salt, Jordan
Sana Janakat
Department of Nutrition and Food Technology, Jordan University of Science and Technology, Irbid, Jordan
Sawsan A. Oran
Department of Biological Sciences, University of Jordan, Amman, Jordan