Research Article
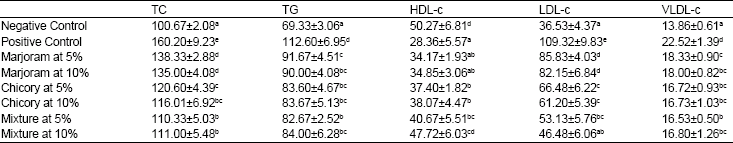
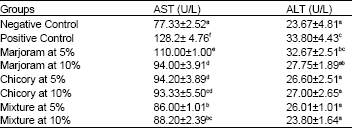
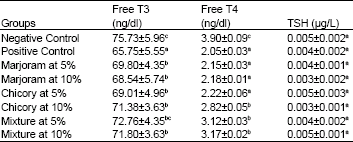
Biochemical and Histopathological Studies on the Water Extracts of Marjoram and Chicory Herbs and Their Mixture In Obese Rats
Department of Nutrition and Food Science, Faculty of Home Economics, Helwan University, Egypt
Reham S. Ramadan
Department of Nutrition and Food Science, Faculty of Home Economics, Helwan University, Egypt
Reham A. Mohamed
Department of Nutrition and Food Science, Faculty of Home Economics, Helwan University, Egypt