Research Article
Assessment of Phytic Acid Levels in Some Local Cereal Grains in Two Districts in the Upper East Region of Ghana
Faculty of Applied Sciences, University for Development Studies, P.O. Box 24, Navrongo, Upper East Region, Ghana
Phytic acid (C6H18O24P6) also known as inositol hexaphosphate (IP6) or phytate as a salt, is the principal storage form of phosphorus in many plant tissues especially bran and seeds. Phosphorus in this form is not bioavailable to non-ruminants because these lack the digestive enzyme phytase, required to separate phosphorus from phytate molecule. On the other hand ruminants readily utilize phytate because they are able to produce the enzyme phytase from their rumen microorganisms.
Normally the salts of phytic acid are found in plant seeds, animals and soils but the acid originates from natural mineral sources containing phosphorus or from fertilizers (Marchner, 1997). As P containing fertilizers are applied to the soils, plant roots pick up the P at a physiological pH mainly as PO-34 which remains as inorganic phosphorus (Pi) and is esterified through the hydroxyl group to the carbon chain (C-O-P) as a single phosphate ester or attached to another phosphate by an energy rich pyrophosphate bond. The rate of exchange between Pi and organic P in the ester and the pyrophosphate bond is very high and this leads to plant roots incorporating Pi into the organic P within a few minutes but this is released almost immediately again into the xylem.
The amount of phytate present in plant seeds and grains ranges from 0.5-5% (Loewus, 2002). It is mainly present as a salt of mono-valent and divalent cations such as K+, Mg2+ and Ca2+. It is accumulated into the seed during the ripening period. In dormant seeds, phytate represents 60-90% of total phosphate. Phytate is regarded as a primary storage form of both phosphate and inositol in plant seeds and grains (Loewus, 2002). It also serves as a store of cations by chelating free metals such as Fe, Zn and a potent anti-oxidant (Reddy et al., 1982).
Phytate is a naturally occurring compound formed during maturation of plant seeds and grains and therefore a common constituent of plant derived food. Depending on the amount of plant derived foods in diets and level of processing, daily intake can be as high as 4500 mg (Reddy, 2002). The mean daily intake of phytate is estimated to be 200-2000 mg for vegetarians and in developing countries and it is about 150-1400 mg. Phytate behaves in a broad pH range as a highly negatively charged ion and has therefore a high affinity for positive charges such as minerals, trace elements and proteins. However, phytate-phosphorus is less nutritionally available since the phytate is not quantitatively hydrolyzable in human gut (Sandberg and Anserson, 1988).
The objective of this work was therefore to undertake a preliminary assessment of the phytic acid levels of selected cereals that are commonly consumed in the northern part of Ghana since these cereals contribute significantly to the nutritional status of the population especially children.
Literature review: Phytic acid is widely distributed in nature because it is a major storage of P in cereals, legumes and oil seeds (Harland Oberleas, 1987). It also occurs in muscles and organs of animals including humans. It is typically found in outer aleouron layer of cereal grains in the endosperm of legumes and oil seeds. The alkali metal (Na, K) salts of phytic acid are usually soluble but those formed with alkali earth and transition metals are practically insoluble (Adu, 1980). It can be synthesized from cyclic alcohol myo-inositol by esterification of the hydroxyl groups with phosphate groups.
The Ca and Mg salts of phytic acid are known as phytin and occur widely in considerable amounts in plants (Adu, 1980). During the development of seeds especially cereals the cells become filled with starch grains and protein bodies known as aleouron which contain phytic acid that accumulates as the seed develops. During germination there is marked increase in the activity of various phosphate uses including phytase. The activity of phytate in cereals is highest in scutulum and aleouron layers which results in the liberation of considerable amount of organic phosphate, calcium, magnesium and potassium for general metabolic processes of the plant (Adu, 1980). However, it has been stated that leafy vegetables, fruits and fermented foods do not contain any phytic acid. Generally the effect of high levels of phytic acid taken in by humans decreases the absorption of most metal ions that are needed for the proper functioning of the body. Among these metals include Cu, Fe, Mg and Ca which have been selected for the study. The fact that phytate complexes at the physiological pH, it is regarded as the main reason why minerals are not available for absorption. Most studies have shown an inverse relationship between phytate content and mineral availability.
Although there are many adverse effects of high intake of phytate in humans, many beneficial effects have also been found. Phytate exerts beneficial effects in the gastrointestinal tract and other target tissues through its chelating ability. It has been demonstrated that phytate as a calcium salt can protect against dietary Pb2+ in experimental animals and human volunteers (Wise and Gilbert, 1981), hence phytate has the ability to counteract acute oral Pb2+ toxicity. Heart disease is a leading cause of death in the Western countries but it is low in Japan and developing countries. Elevated plasma cholesterol or elevated LDL-cholesterol concentrations have been shown to be one of the risk factors. It has been suggested that dietary fibre or more specifically phytate which is a component of fibre can influence the aetiology of heart disease (Potter, 1995). Jariwalla et al. (1990) showed that dietary phytate supplementation resulted in the lowering of serum cholesterol and triglyceride levels. This effect accompanied the decrease in serum Zn level and Zn-Cu ratio. This is because coronary heart disease appears to be caused by an imbalance of Zn-Cu metabolism.
An increase of renal stone incidence in northern Europe, North America and Japan which has been linked with the industrial development of these countries. A study on dietary phytate intake of black and white South Africans indicated that the whites were more affected than the black (Zhou and Erdman, 1995). The dietary difference is that the black population consumes large quantities of food containing high levels of fibre and phytate. However, a high phytate diet has been used to treat hypercalciuria and renal stone formation in humans. Phytate lowers the solubility of calcium, fluoride and phosphate which are the major components of the enamel (Kaufman and Kleinberg, 1971). Thus teeth are protected against the attack of acid and bacteria. Also the high affinity of phytate for hydroxyl apatite may prevent the formation of plaque and tartar. Incidence of cancer especially intestinal cancer has been associated with dietary fat intake which is inversely related to fibre intake. The rate of colonic cancer can arise from many fibre-rich foods which may contain high phytate. Conversely high phytate intake can be a factor in reducing breast and prostate cancer in man (Vucenik and Shamsuddin, 2003).
Location of sample collection: From the literature review it is evident that intake of phytate has both advantages and disadvantages. However, little work has been done on the local cereals which form an important source of diet for the people in Northern Ghana and which have also been demonstrated to be a likely source of phytate for people living in the area. Samples of pearl millet (Pennisetum glaucum), sorghum (Sorghum vulgare) and rice (oryza sativa) were collected from the Kasena-Nankana District (KND) and in the Talensi-Nabdam District (TND) for study in 2007. Samples of soils were collected only at TND between 0-30 cm depth due to lack of logistics. A maximum depth of 30 cm was chosen because most crops have their root zone within that distance. The areas in which samples were collected in the Kasena-Nankana were Gnani, Gongnia for sorghum and pearl millet while rice was obtained from the Irrigation Company of Upper Region (ICOUR) project site. In KND parameters analyzed were Cu, Fe, Mn and Zn while those analyzed at TND were Cu, Fe, Ni and Zn. Phytic acid in the cereals was analyzed in both districts. However, for the soil samples from TND, pH, Co, Fe, Zn and Cation Exchange Capacity (CEC) were analyzed. The elements analyzed in both districts were not exactly the same. The focus was to determine the level of phytate in the cereals from the two areas.
Samples of cereals were first air-dried, cleaned, ground to powder and sieved using a 150 um sieve. About 0.5 g of the powdered sample was put into a beaker followed by 4 ml HNO3 with constant stirring until after 10 min when it was now filtered and transferred into a 100 ml volumetric flask. It was then made up to the mark with distilled water.
Soil samples were also air-dried to expel all moisture ground with mortar and pestle to very fine particles that could pass through 150 mm. About 0.2 g of soil sample was weighed into a beaker and 4 ml aqua-regia (3 ml, HNO3 and 1 ml HCl) was added. The mixture was stirred and put on a water bath for about an hour. After this it was removed, cooled and filtered twice using 150 mm diameter filter paper. The filtrate was transferred into 100 ml volumetric flask and made up to the mark. All the filtrates were sent to Water Research Institute (WRI) of the Council for Scientific and Industrial Research (CSIR), Accra, Ghana for analysis using Unicam 969 atomic absorption spectrophotometer.
For the analyses of phytic acid, 2 g of cereal was put in an Erlenmeyer flask and 25 ml of 3% HCI added. This was allowed to stand for about 45 min and then centrifuged. Then 5 ml of the supernatant solution was taken and 2 ml of FeCl3 solution added. This was heated for about 25 min and centrifuged again to obtain the ferric phytate precipitate. The precipitate was then washed with 3% HCI and later dispersed in water and 2 ml of 1.5 M NaOH and made up to a volume of 15 ml with water. It was then filtered and washed again with 3.2 M HNO3 and put into a 100 ml volumetric flask made up to the mark. Another 5 ml portion was transferred into another 100 ml volumetric flask and brought up to 70 ml. Then 20 ml of 1.5 M potassium thiocyanate was added and the concentration of Fe3+ obtained using a UV spectrophotometer. These results obtained for Fe3+ were used to estimate the amount of phytate phosphorus assuming 4Fe:6P. For the soil samples, the pH was obtained by adding 20 g of soil to 40 ml of distilled water and stirring for some time. Before taking the pH reading the instrument was calibrated with standard solutions of pH 4, 7 and 10.
RESULTS
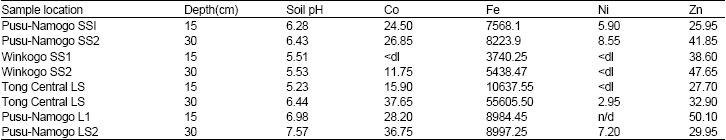
Concentration of metal elements in soils: The results of samples of soils are listed in Table 1. The pH of the top soil was generally lower than the second horizon.
In Pusu-Namogo where two types of soils were analyzed for pH, it was observed that the loamy soils had higher pH than the sandy soils from the same area. The highest pH values were obtained from Pusu-Namogo which ranged from 6.28-7.57. while the soils in Winkogo were the most acidic (pH = 5.51-5.53) even though Tongo Central top loamy soil had the least pH = 5.23. The trend of concentration of the metals was similar to that observed in the pH. All the soils had the metal concentrations higher at the lower horizons. Concentration of Fe was very high in the samples than any other metal. Ni concentration was either low or below detection. This could be due to the inherent low levels of Ni in the bedrock in the area. On the other hand, Fe is a major element of the earth crust and was found to be high in all the soil samples.
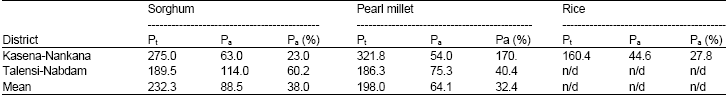
Levels of P in soils and cereal: Total phosphorus (Pt) and that associated with phytic acid (Pa) were significantly high in all the cereals. Total phosphorus of pearl millet and sorghum ranged from 118-254.5 while phytic acid phosphorus Pa ranged from 59.25- 114.25 mg/100g resulting in a percentage range of 23.3- 64.3% in the Talensi-Nabdam District. In the Kasena-Nankana District, Pt ranged from 176.5-467 mg/100g while that due to Pa was 48.0-72.0 mg/100g. Total phosphorus in rice was comparatively high and ranged from 71.5-316.5 while the phytic acid phosphorus also ranged from 22.95-103 mg/100g.
Generally the values of Pt and Pa indicate a relatively higher concentration of P in all the cereals including rice in the two districts. This is shown by the high mean values, that of sorghum and pearl millet were respectively 275.0 and 321.7 mg/100g in KND while in TND, the respective mean values were 189.5 and 186.5 mg/100g. The rice had mean total phosphorus of 160.4 mg/100g (Table 2). The trend of phosphorus concentration derived from phytic acid was observed to be opposite to that of total phosphorus in the two districts. While Pt was considerably higher in KND, the Pa was higher in TND, which suggests that the percentage of phytic acid in TND will be higher, although rice had a significantly high Pt, the Pa was comparatively lower. It was observed that sorghum and pearl millet in the TND area had comparatively lower Pt in the two cereals but higher Pa than that in KND. In the two areas the mean Pt and Pa values were below the amount found in dormant seeds which is 60-90% as suggested by Loewus (2002) but higher than fresh seeds. Only sorghum in the TND was above 60% Pa. The cereals from KND although had considerable amount of Pa yet all were below that of dormant seeds. This indicates that either the seeds were fresh and had not undergone some dormancy to allow the development of phytic acid or they were inherently lower in phytic acid. This could require further investigation.
The relatively low level of phytic acid in these cereals suggests that many of the trace metals that are required for proper functioning of the body will not be precipitated by the acid and thus will be available in nutrition.
Rice is a common cereal found in every home and consumed by many people and with its relatively low level of Pa suggests that the needed nutrients in terms of trace metals can be available to them. Pearl millet is commonly used for the local porridge, which is one of the cheapest meals for breakfast and if it should contain high phytic acid, its use may be restricted which can economically affect people who need additional supplements of trace metals.
Concentration of metal elements and phytic acid in cereals: Concentration of metals and percentage phytic acid for pearl millet in the Talensi-Nabdam and that for Kasena-Nankana Districts are presented in Table 3.
| Table 1: | Levels of selected chemical elements (in mg/L) and pH in soil samples. (dl = less then detection limit) |
 | |
| Table 2: | Mean levels of total (Pt) and phytic acid derived (Pa) Phosphorus and n/d = not determined |
 | |
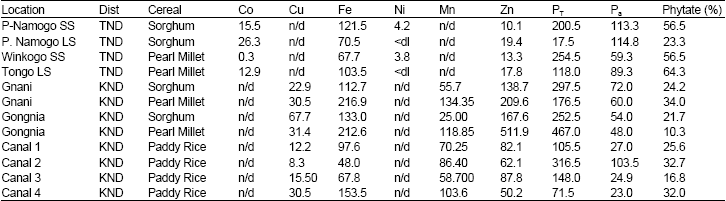
The Fe and Zn metals were analyzed in both districts and the results indicated high concentrations in all the cereals and at all sites.
Iron concentration ranged from 67.80 in paddy rice to 216.85 mg/kg in pearl millet at Gnani in the Kasena-Nankana District while Zn concentration ranged from 10.10 from Pusu- Namogo in TND to 511.85 mg/kg in Gongnia in KND. However, concentrations of Fe and Zn from the Kasena-Nankana were relatively higher than the corresponding cereals from Talensi-Nabdam. Furthermore, pearl millet from the two locations in Kasena-Nankana District (Gnani and Gongnia) had higher levels of Fe and Zn than both rice and sorghum. Levels of phytate ranged from 10.3-75.6%.
The highest phytate was obtained from sorghum from Pusu-Namogo in the Talensi-Nabdam District while the lowest was from the pearl millet but from Gongnia in the Kasena-Nankan District. The paddy rice had a close range of phytate from 16.82-32% with a mean value of 26.8% as compared to the other cereals of 36.2 and 41.3% for pearl millet and sorghum respectively. This suggests that paddy rice had less phosphorus than the other two cereals. The mean levels of the various cereals are presented in the Table 4. Phytic acid levels were higher in all the cereals from the Talensi-Nabdam area than those in the Kasena-Nankana probably due to longer maturation or absorption from inorganic fertilizers. This explains why the trace metal content was relatively higher in KND than those in TND. The high levels of Pa could be due to the application of artificial fertilizers. However, the cereals from KND had the lowest phytate level but also recorded the highest Fe and Zn levels.
Regression analyses performed on the samples for various metals (Cu, Fe, Mn and Zn) and phytic acid showed that sorghum had the least linear R2 values (Fig. 1) with Fe and Zn. This suggests that phytic and phosphorus and the meals are not derived from the same sources.
Pearl millet on other hand had R2 values for phytic acid against Fe and Zn to be 0.73 and 0.96 respectively (Fig. 1) suggesting a high association between them but the association was inversely related suggesting that increased phytic acid level led to a decrease of the metal concentration.
For the paddy rice, Fe and Cu had R2 values less than 1%. Zinc and Mn had R2 values greater than 0.78 and 0.79 respectively. However, Zn had an inverse association with phytic acid while the R2 for Mn was directly associated.
DISCUSSION
Levels of metals in the soils: The soil pH range (5.23-7.68) was from slightly acidic to weakly alkaline and most fell within the physiological pH range of 6.5-8.0 (Mathews et al., 2000) suggesting that any available phosphorus can be absorbed from the soil medium. The slight acidity also enhanced a suitable medium for many rock minerals to dissolve and release metal ions such as Co, Fe and Zn. Low values of Ni in some samples either reflected the inherent levels in the parent material or that the geochemical conditions were not suitable for the release of the metal from its parent minerals. Most fertilizers do not contain high Ni where it could serve as an important source of the element. A high level of Fe was however attributed to its geological abundance in the earth crust and that the pH range and other geochemical conditions especially in TND favoured its release into the environment. Phytic acid and selected metals in cereals.
The mean phytic acid obtained from this work was higher in pearl millet than sorghum and rice. This is in line with what had earlier been found by Adu (1980) who earlier found that phytic acid in millet was higher than sorghum and rice. These two cereals usually take different periods of maturation before harvesting. The sorghum takes relatively shorter period of 2-3 months while the pearl millet takes a longer time of about 4-5 months.
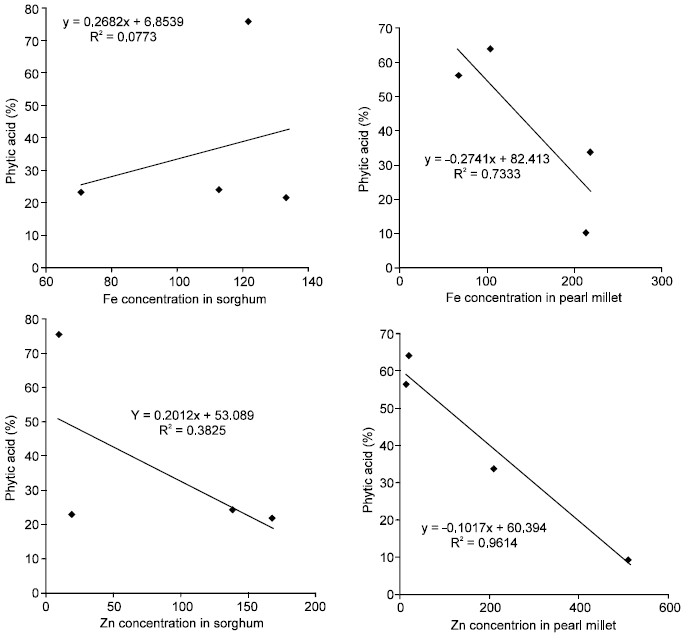 | |
| Fig. 1: | Plots of concentration of metals with phytic acid (%) |
It may be suggested that the longer the period the more absorption of P the plant takes and stores which explains the difference between the pearl millet and the other two plants. Paddy rice takes a much shorter period for maturation since there are normally multiple harvests in the year. Moreover, due to high water content demanded by rice any soluble P can infiltrate with water below the root level of the rice plants and hence less P is available for absorption.
The mean level of phytic acid obtained from cereals was less than the range quoted in literature which is 60-90% (Loewus, 2002) and that obtained by Adu (1980). This suggests that these cereals are generally low in phytic acid and therefore low P. This implies that more cations are not bound to the phytic acid molecule hence can be bioavailable for absorption. Human beings do not have the enzyme phytase to be able to metabolize phytic acid for nutritional purposes. In such cases, trace metals normally required in minute quantities can be made available for the body. The second implication is that P which is required for bone and other hard parts of the body may not be sufficient unless there is supplementation from other sources.
Phytic acid in rice and sorghum were found to be unrelated with Fe and Cu (Fig. 2). It could be explained the source that the absorbed metals and phytic acid were not derived from the same source. Furthermore the soil structural differences could also affect the absorption of the metals since very lose soil will not be able retain the solution carrying the metals for enough time for the process to occur. For example concentration of Fe in Pusu-Namogo sand and loamy soils produced different levels of phytic acid and Fe. Higher levels of phytic acid were obtained from sorghum grown from sandy soils than from the loamy soils containing finer particles.
| Table 3: | Metal elements and phytate levels in cereals from Talensi-Nabdam District (TND) and Kasena-Nankan District (KN D) of Upper East Region, (Note: n/d= not determined ’ <dl= less than detection limit, PT = total P and Pa due to phytic acid |
 | |
| Table 4: | Mean levels of phytic acid and selected metals in cereals |
 | |
This means that the finer the soil particles the less the amount of phosphorus available for absorption by plant from loamy soils. This is because P is normally absorbed on fine particles and thus become less available in the soil medium for plant absorption (Kabata-Pendias and Pendias, 1992). The third reason is attributed to geological differences. Although the Kasena-Nankana and the Talensi-nabdam Districts lie within the same climatic and vegetational zones, there may be some micro-environmental differences in soil types and chemical composition arising from the type of rock and process of weathering. Areas with more sandy soils as may be the case of the Talensi-Nabdam District enhances water infiltration and will not impede the movement of free P which will become available for absorption by plants than silty loamy soils as the case for Kasina-Nankana.
The regression coefficients for phytic acid with Fe and Zn respectively for pearl millet were high and negative suggesting an inverse relationship. The R2 values of Co, Cu and Ni with phytic acid were low. These values suggested that the trace metals and phytic acid were not derived from the same source. This is because phytate ion has six negative charges per molecule for which can be used to attract six positive charges. Different positive ions will therefore competition for negative sites. If all the negative sites of phytate ion sites are occupied, the excess metal ions will remain uncombined. Chelation or precipitation will not take place again until there are more negative sites produced by addition of phyate ions. Chelation depends on the charge density which is function of ionic radius and the amount of charge. Highly charged ions with low ionic radii have high charge densities and will chelate faster with phytic acid. Iron exist in most soils as Fe3+ and has an ionic radius of.74 nm while Co2+, Cu2+ and Ni2+ in their most stable forms have radii of 0.73, 0.74 and 0.70 nm (Sharpe, 1992), Co2+ has a similar radius with Fe3+ but slightly lower for Cu2+ and Ni2+ and lower charge. Hence Fe3+ will chelate faster due to higher charge density and this explains why Fe3+ will combine with phytic acid faster and more than the others. The low concentration of the other ions (Co2+, Cu2+ and Ni2+) could be attributed to a reduction by chelation with phytic acid which subsequently has some implication to nutrition. Free metal ions will be bioavailable for animals including humans who do not have the enzyme phytase to assist in digesting phytin to release the bound metals for embolic purposes.
Nutritional benefits of phytic acid in cereals: It has been stated that intake of high phytate-containing food substances provides numerous benefits such as the prevention of breast cancer and prostate cancer, lead poisoning, heart disease and renal stone formation (Vucenik and Shamsuddin, 2003).
Pearl millet is usually the preferred cereal for the morning porridge not only in Northern Ghana but the whole country, which many people use for breakfast. Sometimes sorghum is used. Paddy rice is used in most households and for the preparation of the common ‘rice and beans ’ (locally known as “waache ”) has some level of phytic acid. High phytic acid content may prevent many people getting adequate nutrition since any phytic acid may interfere with the intake of trace metals that are required in small amounts by the body. For the supply of adequate amount of phytic acid containing diet some of the cereals need to be preserved and cultivated in some reasonable quantities. Cobalt, Ni and Zn have been found to be linked with malnutrition cereals containing phytic acid will further reduce their levels thus worsening the negative impact of these metals in humans and even other animal consumption.
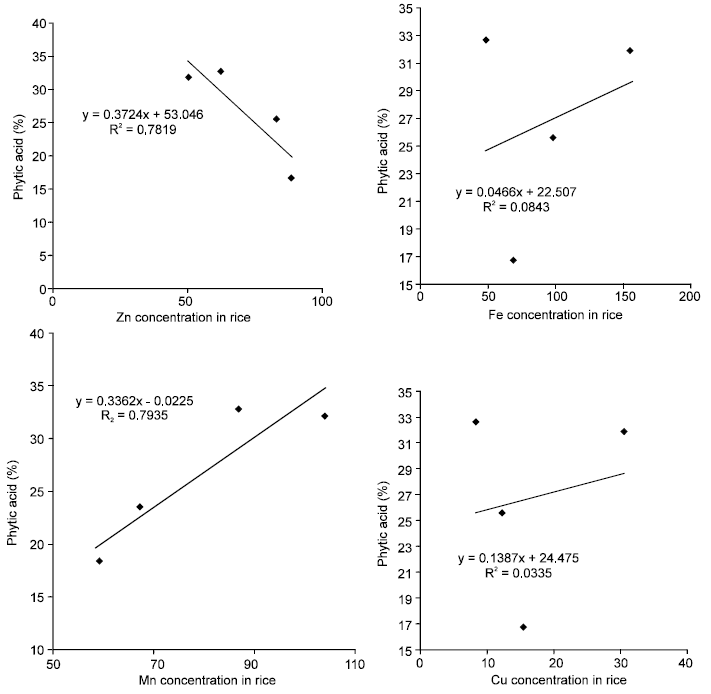 | |
| Fig. 2: | Scatter plots of concentration of metals with phytic acid (%) |
However, the benefits of phytic in food substances are enormous and therefore cannot be eliminated.
Conclusion and recommendation: The study has shown that sorghum, pearl millet and rice contain phytic acid of varying levels. Sorghum was found to contain high total P in the Talensi-Nabdam District while pearl recorded a higher value in the Kasena-Nankana District. Phytic acid phosphorus was highest in pearl millet from TND all lowest in the same cereal in Gongnia from the Kasena-Nankana District. However, it was observed that rice on the average had the least amount of phytic acid. Selected trace metals analyzed showed varying levels. In rice Co and Ni could not be detected but this could not be ascertained whether the low levels was due to precipitation with phytic acid. Regression did not indicate any relationship between some of the metals (Fe and Zn) concentrations and phytic acid for rice and sorghum and no relationship was found with the phytic acid level with Cu. However, Zn and Fe showed relatively high regression coefficients for pearl millet and showing an inverse trend suggesting that the higher the amount of metal concentration the lower level of phytic acid in the cereal.
This work has given insight to the amount of phytic acid levels in a few selected cereals that are often used by the population since these form the major food sources. High phytic acid in cereals can lead to essential elements deficiency and hence cause malnutrition. Phytic acid also has positive effects in human health. It is therefore necessary to have knowledge of phytic acid levels for cereals that are produced locally and also link its level to phosphorus status of the soils where the cereals are produced.
I wish to acknowledge the contributions and works of the following students who worked tirelessly to produce the preliminary results for confirmation. These students are Mr Mahanmadu Tia and Mr Louis Nyamordey. Furthermore, all those assisted in proof reading the draft of the manuscript before it was sent to the press are duly acknowledged.