Research Article
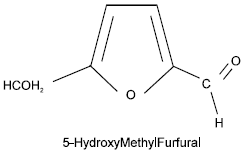
Identification and Quantification of 5-Hydroxymethyl Furfural HMF in Some Sugar-Containing Food Products by HPLC
Central Laboratory, Ministry of Science and Technology, Khartoum, Sudan
Mohammed Idrees Taha
Central Laboratory, Ministry of Science and Technology, Khartoum, Sudan
Badawi Ahmed Zakaria
Central Laboratory, Ministry of Science and Technology, Khartoum, Sudan
Babeker Siddig
Central Laboratory, Ministry of Science and Technology, Khartoum, Sudan
Hazeim Mahmod
Central Laboratory, Ministry of Science and Technology, Khartoum, Sudan
Abedel Rahim Mohamed Elhussein
Central Laboratory, Ministry of Science and Technology, Khartoum, Sudan
Elrasheed Ahmed Gad kariem
Ribat University, Khartoum, Sudan