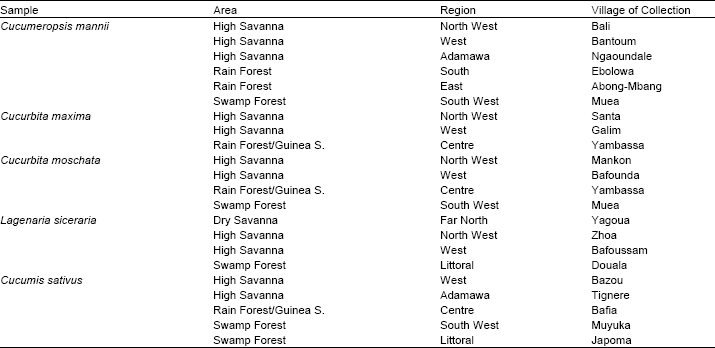
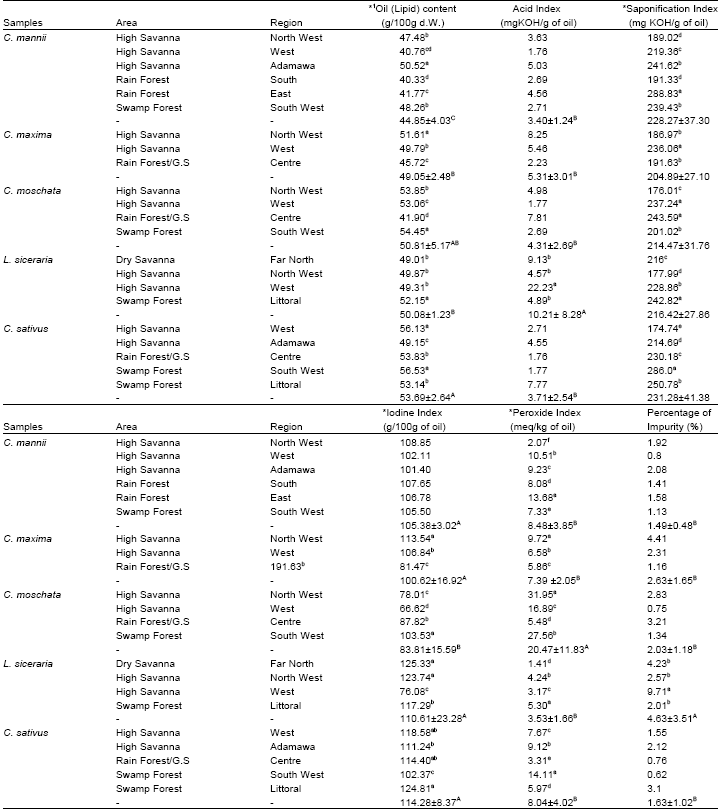
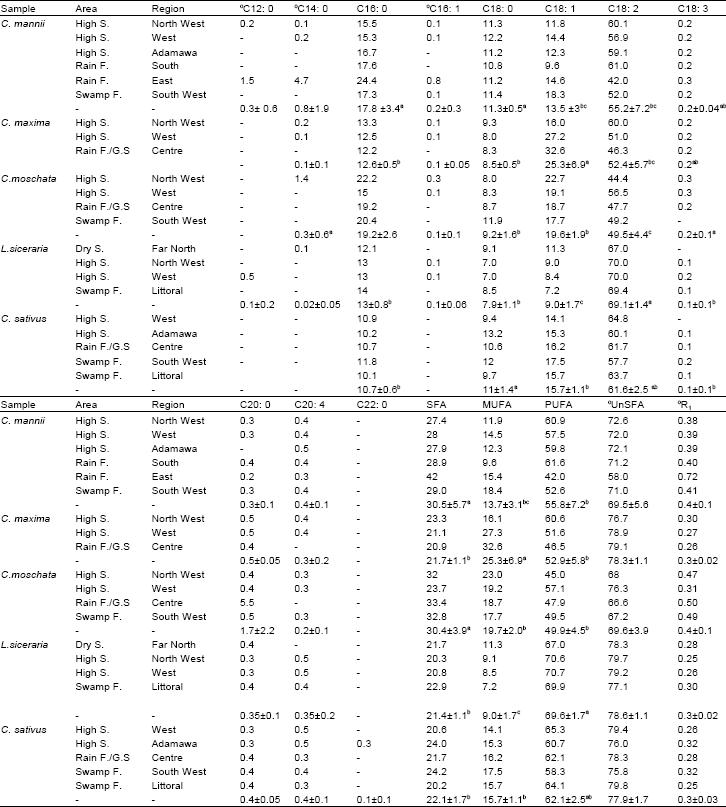
Research Article
Chemical Properties of Some Cucurbitaceae Oils from Cameroon
Department of Biochemistry, Faculty of Science, University of Yaounde I, P.O. Box 812, Cameroon
M.B. Achu
Department of Biochemistry, Faculty of Science, University of Yaounde I, P.O. Box 812, Cameroon
G. Kansci
Department of Biochemistry, Faculty of Science, University of Yaounde I, P.O. Box 812, Cameroon
R. Ponka
Department of Biochemistry, Faculty of Science, University of Yaounde I, P.O. Box 812, Cameroon
M. Fotso
Centre for Research in Food and Nutrition, IMPM, Yaounde, Cameroon
C. Tchiegang
Department of Food Science and Nutrition, ENSAI/IUT, Ngaoundere, Cameroon
F.M. Tchouanguep
Department of Biochemistry, Faculty of Science, University of Dschang, Cameroon