Research Article
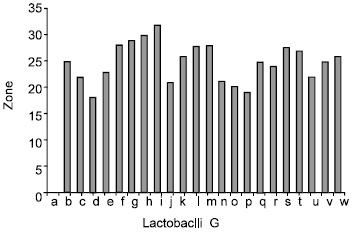
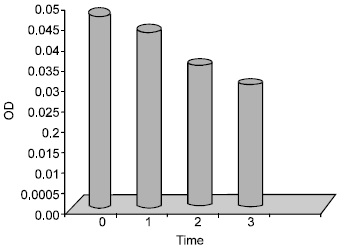
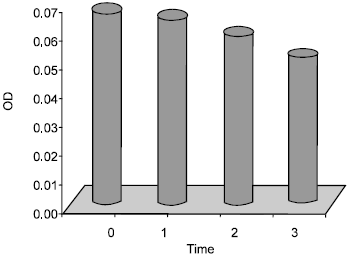
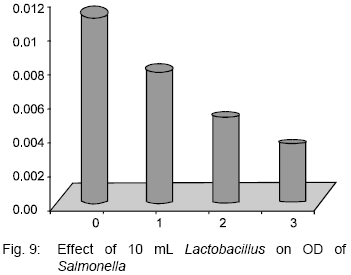
Antibacterial Effect of Authochlorous Lactobacillus Strains Isolated from Traditional Yogurts
Department of Microbiology, Faculty of Medicine, Medical Sciences, University of Isfahan, Isfahann, Iran
Nazila Hassannia
Department of Microbiology, Faculty of Medicine, Medical Sciences, University of Isfahan, Isfahann, Iran
Mohammad Reza Fazeli
Department of Microbiology, Faculty of Medicine, Medical Sciences, University of Isfahan, Isfahann, Iran
Asghar Havaee
Department of Microbiology, Faculty of Medicine, Medical Sciences, University of Isfahan, Isfahann, Iran
Hossein Jamalifar
Department of Drug and Food Control, Faculty of Pharmacy, Medical Sciences, University of Tehran, Tehran, Iran
Mearaj Pour Hossein
Department of Microbiology, Faculty of Medicine, Medical Sciences, University of Isfahan, Isfahann, Iran
Hassan Shojaee
Department of Microbiology, Faculty of Medicine, Medical Sciences, University of Isfahan, Isfahann, Iran
Mojtaba Akbari
Department of Microbiology, Faculty of Medicine, Medical Sciences, University of Isfahan, Isfahann, Iran