Research Article
Probiotics Bacteria in Fermented Dairy Products
AL-Balqa Applied University, Irbid University College, Jordan
Ayman Suliman Mazahreh
Al-Balqa Applied University, Princes Alia University College, P.O. Box: 941941, Amman 11194, Jordan
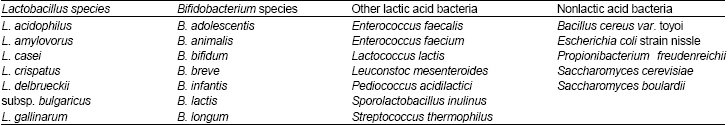
The growing popularity of yogurt over the years, has largely been increased due to its perceived health benefits. Yogurt is one of the best-fermented dairy food that contain "probiotics" which is a living microorganism, upon ingestion in sufficient amount, exerts beneficial effects on the normal microbial population of the gastrointestinal tract (Bourlioux et al., 2003).The most commonly defined probiotic bacteria include member of the genera Lactobacillus, Bifidobacterium, lactic acid and non lactic acid bacteria. Different species belong to these genera are considered as probiotics (Table 2). Many fermented dairy products, were supplemented with variable probiotic strains, like Lactobacilli, Streptococcus and Bifidobacterium strains (Table 1 and 2).
Yogurt is a coagulated milk product, which results from the fermentation of lactic acid in milk, by probiotics Lactobacillus bulgaricus and Streptococcus thermophilus (Pelczar et al., 1986). It's traditionally manufacture by fortifying whole or skimmed milk by evaporation, or addition of skim milk powder, heating to 85-95oC for 10-30 min, inoculating with probiotic bacteria and then incubating at 42-45oC. Bifidus yogurt prepared, by the addition of B. bifidum and B. longum, to the yogurt culture and incubating at 42oC for 3-4 h. L. Acidophilus can also incorporate with yogurt culture to create acidophilus yogurt. National Yogurt Association Rules (NYAR) define the active culture yogurt as a final product that contains live probiotic bacteria in amount = 108 cells/ g at the end time of the manufacture (Mazza, 1998).
Dairy products such as milk, cheese and yogurt have been recognized as excellent sources of vitamins and minerals including riboflavin, phosphorus and calcium (Mazza, 1998). Prebiotics are non digestible food ingredients that beneficially affect the host by stimulating the growth and the activity of colonic microbiota. Non digestible oligosaccharides and fructo-oligosaccharides are considered as probiotics. They have been shown to stimulate the growth of endogenous Bifidobacteria, which after a short feeding period, become predominant in human faces. Also it can modulate lipid metabolism via fermentation product. Probiotics and prebiotics modulate the composition of the gut microbiota especially in the colon. The beneficial effects may result from suppression of pathogens and the stimulation of probiotics growth that contributes in a positive way to the nutrition and health of the gut (Naidu et al., 1999). The present study focuses on the effect of fermented dairy product consumption as a basic Mediterranean dietary tradition, improving the health and longevity. Among adults, life expectancy was the highest in the world and the rate of coronary heart disease, cancer and other diet related chronic disease, were among the lowest, in which yogurt is considered the main source of fermented diary products in the Mediterranean diet, incorporated with other healthy foods (fruit, vegetable, L. gasseri L. johnsonii L. paracasei L. plantarum L. reuteri L. rhamnosus brown bread, cereals and olive oil) (Willett et al., 1995).
| Table 1: | Lactobacillus strains used in probiotic yogurts or yogurt-like products |
 | |
| Table 2: | Microorganisms considered as probiotics |
 | |
Even recent trends of probiotics are based on expectations of prophylactic effects and as alternative to some pharmaceutical preparations (Sanders, 1999). This study aimed to prove the beneficial therapeutic effects of different probiotics culture on the gut function, involves the enhancement of the immune response against gastrointestinal disorders. However, results have been inconsistent, which may be due to the differences in probiotic strains, routes of administration, or types of procedure used in these studies.
Historical background: The concept of probiotics evolved at the turn of the 20th century, from a hypothesis first proposed by Nobel Prize winning Russian scientist Elie Metchnikoff, who suggested that long, healthy life of Bulgarians, resulted from their consumption of fermented milk products provided with probiotics. He believed that consuming the fermenting (lactobacillus) positively influenced the microflora of the colon, as well for deceasing the toxic activity of pathogens (Bibel, 1988). The major strains Lactobacillus acidophilus, L. casei and various Bifidobacterium species, B. longum, B. bifidum are the most dominant species in human small and large intestine, that can inhibit the growth of pathogenic organisms, through production of organic acids and bacteriocins (Mazza, 1998). Up to date, many research studies focused on the role of probiotic bacteria in the yogurt for the enhancement of the gastrointestinal function through increasing minerals absorption, reduction in lactose intolerance (lactase deficiency) (Vesa et al., 2000, Martini et al., 1991). The consumption of L. acidophilus and S. thermopillus culture in yogurt enables the inhibition of the Pathogen infection through production of acetic acid, lactic acid and bacteriocins (Bianchi-Salvadori, 1986), also for synthesis of other compounds like peroxide, pyroglutamate by Lactobacilli strains improves the inhibition of pathogens (Silva et al., 1987). As well for stabilizing the intestinal microflora after long term antibiotics uses (Brown et al., 2005). Suppression of the harmful carcinogens associated with colon cancer (Tavan et al., 2002; Wollowski et al., 1999) and increasing the immune response by production of secretory immunoglobulin was achieved (Perdigon, 1995, 2003). Relief of constipation, reduction of serum Cholesterol (Jones, 2002), allergy in young adults (Piaia et al., 2003) and control of diarrhea was established through the consumption of yogurt (Heyman, 2000).
Physiological properties of probiotics: Probiotics including lactic acid bacteria are gram positive, non- spore, catalase-negative bacteria characterized as fastidious, acid-tolerant and fermentative microorganisms. Its can inhabitant human oral cavity, vagina and gastrointestinal tract, through which Lactobacillus acidophilus, L. johnsonii and L. casei groups are the most associated to the intestinal tract (Holzapfel et al., 2001). The functional properties and safety of probiotics strains are mentioned in (Table 3).
Furthermore Bafidobacteria constitute most of the flora of breastfed infants, able to survive the intestinal digestion and appear in stool. The main species inhabit the infant intestinal tract are B. bifidum, B. infantis, B. breve and B. longum. They are anaerobic, non motile, gram positive curved rods, which produce lactic acid and acetic acid upon fermentation of the milk, able to reduce the PH of the intestine, which in turn limits the growth of pathogens and other putrefactive bacteria. Prebiotics non digestible oligosaccharides are complex carbo-hydrates, able to resist hydrolysis by salivary and intestinal digestive enzymes and stimulate the growth of bifidus flora in the gut (Naidu et al., 1999).
| Table 3: | Successful probiotic strains and their functional properties1 |
 | |
Probiotics and nutrition value of dairy products: Nutritional value of the final diary products depend on the milk-based sources (mammalian type, feed, age and lactation stage) and processing factors (temperature, heat duration and storage condition). Also the type of probiotic strains used in the fermentation, directly influences the nutritional and physiological value of the final yogurt product (Mazza, 1998).
Vitamins: Dairy products are considered a major source of vitamins, including vitamin B-6, Vitamin B-12, riboflavin, niacin and folic acid. Folate is one of the B vitamins that some probiotics can synthesize (Kneifel et al., 1992). Various bacterial species were also used for milk fermentation and yogurt production was examined for its ability to synthesize or utilize folate. S. thermophilus and Bifidobacteria were folate producer, while Lactobacilli depleted folate from the milk media. Fermentation using combination of Bifidobacterium animalis and S. thermophilus result in six- fold increase in folate concentration (Crittenden et al., 2003).
PH: The acidic PH of yogurt ionizes calcium and thus facilitates the absorption of calcium in the intestine (Bronner and Pansu, 1999). Low PH of yogurt reduces the inhibitory effect of phytic acid on calcium bioavailability. Vitamin- D plays a major regulatory role in intestinal calcium absorption. The active transport of calcium absorption in duodenum and proximal jejunum requires calbindin-D, vitamin D-dependent calcium-binding Protein (Norman, 1990). Diary products such as milk and infant formula are fortified with vitamin D, with 2.5 μg (100 IU) Vitamin D/237-ml, unlike yogurt that is not fortified with vitamin D.
Lactose: Dairy products are considered one of the sources of disaccharide lactose in human diets. Before absorption, lactose is hydrolyzed by the intestinal brush border β-galactosidase (lactase) into glucose and galactose. Probiotics that present in yogurt, Such as L. bulgaricus and S. thermophilus, expressed functional lactase that can hydrolyses 20-30% of the lactose, which contributes to better tolerance of lactose in yogurt than that of milk, by person with lactose maldigestion (Vesa et al., 2000 and Martini et al., 1991).
Health aspect of probiotics on the gastrointestinal function
Probiotic interaction: Yogurt and probiotics bacteria contribute to several factors that enhance the gut function and health: the make of gastrointestinal flora, the immune response against pathogens. Gut microflora plays a major role against exogenous infectious bacteria through colonization resistance. Most of the bacteria that cross the barriers of stomach and small intestine will be live, metabolically active and colonized with in the gut ecosystem (Bourlioux et al., 2003). Some of these bacteria are useful to the host health, such as Bifidobacterium and Lactobacillus, which are the most dominant genera in the intestinal tract, others are harmful and cause disease to the host. Selection of strain of Bifidobacteria (B. infantis, B. bifidum, B. adolescentis and B. longum) as a probiotic (Table 3), to be included in fermented milks, has been based on the survival in the final product and through the intestinal tract (Holzapfel et al., 2001). B. animalis and B. adolescentis were reported to have a high survival rate during intestinal transit, able to adhere to the receptors of epithelial cells of small and large intestine and induce lisosomal activation of the epithelial enterocytes (Perdigon et al., 2003). The ability of probiotics bacteria to bind to the intestinal brush border tissue, compete with the pathogen to prevent them from accessing the gastrointestinal mucosa (Bernet et al., 1994). Such binding, is influenced by certain adhesion sites with in the intestinal lumen for the bacteria to be contact. These sites are genetically controlled by the host (Bourlioux et al., 2003). LAB bacteria can adapt the host intestine and survive against gastric PH, digestive enzyme and bile Salt (Alm and Pettersson, 1980).
Probiotics differ in their ability to survive in the gastrointestinal tract. In the group of elderly patients with atrophic-gastritis and hypochlorhydria, Lactobacillus gasseri survived passage through the gastrointestinal tract, but S. thermophilus and L. bulgaricus were not recovered (Pedrosa et al., 1995). L.casei present in the fermented diary product, able to survive in the intestine of human flora-associated mouse model and initiate new protein synthesis during its transit with the diet, that influence human health related to that protein (Oozeer et al., 2002). Association of L. casei with yogurt starters probiotics (lactic acid) bacteria in the fermented milk, increase the activity of glycolytic enzyme β-galactosidase, improved for lactose digestion, while for α-and β-glucosidase, that needed for the fermentation of resistant starch, which lead to butyrate production, that improved bowel habits and increase stool output (Djouzi et al., 1997).
Pathogens control: The ability of probiotics bacteria to avoid the gastrointestinal invasion of pathogenic microorganisms has been reported. L. acidophilus-mediated inhibition of the adherence and the entry of enteropathogenic Escherichia coli and Salmonella typhimurium to the enterocyte cell-line Caco-2 cells (Bernet et al., 1994). Also the similar inhibitory effects for two strain of Bifidobacteria (B. breve and B. infantis), were observed (Bernat et al., 1993). The important defensive line of the intestine is based on three essential constituent; gut microflora, mucosal barrier and the innate immune system gut- associated mucosal lymphoid tissue (GALT). GALT is divided into inductive and effector sites. The inductive sites are in the Peyers patches (aggregate glands), which consist of large lymphoid follicles in the terminal small intestine. Specialized transport cells (M cells) and dendrite cells (macrophages) which were found in the epithelial layer of the patches, can phogocytose soluble antigens and pathogens bind to such layer (Bourlioux et al., 2003). The gut microflora (Bifidobacteria and Lactobacilli) plays an important role into protecting the mucosal surface from pathogens, avoiding their attachment and entry into the intestinal mucosa (Perdigon et al., 2003). Also the interaction of probiotics with the mucosal epithelial lining of the gastrointestinal tract, as well as with the lymphoid cells residing in the gut, have been suggested as the most important mechanism by which probiotics enhance the gut immune response against ingested pathogens (Bourlioux et al., 2003). Sixteen strains of Lactobacillus isolated from humans, mice and food product were screened for their capacity to associate with Peyers patches in mice. Lactobacillus fermentum exhibits a preferential binding to the follicle-associated epithelium of the Peyers patches (plant and Conway, 2001). As well for Bifidobacterium animalis which demonstrated by fluorescent labeling techniques, able to interact with the immune cells of Peyers patches of small intestine and large intestine (Perdigon et al., 2003). The effecter component of the mucosal immune system is the secretory immunoglobulin (sIgA).It inhibits the colonization of pathogenic bacteria in the gut, as well as the mucosal penetration of pathogenic antigen.
Immune modulator: Many studies focused on the role of probiotics bacteria for modulating IgA concentration in the gut. Orally administered L.acidophilus and L. casei and the feeding of yogurt increased both (IgA) production and the number of lamina propria B cells that secrete (IgA) in the small intestine of mice. Yogurt is able to inhibit the growth of the intestinal carcinoma by increasing the activity of (IgA, T cells and macrophages) (Perdigon et al., 1995). Modulation of cytokine production by probiotics with in yogurt has been investigated. In addition to interleukin (IL)-1β and tumor necrosis factor (TNF) α, which are produced by macrophages. T lymphocytes (Th1) and (Th2) helper cells, upon activation were able to produce 2 patterns of cytokines (Mosmann et al., 1986). Th1 cells produce interferon-γ (IFN-γ) and IL-2. IFN-γ improves induction of other cytokines and in mediation of macrophage and natural killer cell activation. Th2 cells augment humoral immunity through production of IL-4, IL-5, IL-6 and IL-10 cytokines. The production of IFN-γ in vitro culture using human lymphocytes was reported to be greater in the presence of probiotics (L. bulgaricus and L. thermophilus). The most important target of probiotics mechanisms which affect the gastrointestinal function, including those that control transit time, bowel transit and mucosal motility, as well as those that modulate epithelial cell proliferation, balancing colonic microflora and influencing gastrointestinal immune system. Combination of probiotics and prebiotics within diary products, improves growth and the survival of gut microflora Bifidobacteria and Lactobacilli, influence their effect in the large bowel (Roberfroid, 2000). Combination of two probiotic strains L. gasseri and L. coryniformis in the diary fermented product, instead of L. bulgaricus yogurt strain, were able to survive in the intestine and recorded in high level in the feces of volunteers. Orally intake of probiotic strains increases the concentration of fecal lactic acid bacteria, thus improving the fecal moisture, frequency and the volume of the stool (Olivares et al., 2006).
Clinical application of probiotics
Lactose intolerance: Lactose intolerance involves a reduction of lactose activity in the intestinal brush border of mammals as they age after weaning. The ingestion of dairy Products containing lactose leads to symptoms of lactose intolerance, such as bloating flatus, abdominal pain, abnormal gas and diarrhea (Shermak et al., 1995). Lactose in yogurt is better digested than lactose in other dairy food by lactose intolerance individuals, due to the intra-intestinal activity of the probiotic β-galactosidase (Martini et al., 1991). Most of probiotics bacteria which are used in milk fermentation, including L. bulgaricus and S. thermophilus, can exert their lactase activity in vivo in the gut lumen of both adults and children, thus facilitating digestion and alleviating intolerance (Shermak et al., 1995). L. acidophilus able to modify the lactose fermentation, by human colonic microflora in vitro in lactose maldigesters.
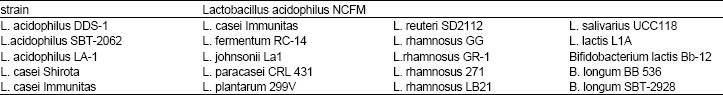
| Table 4: | List of probiotic strains used in commercial manufacture products |
 | |
Diarrhea disease: Microbial balance is an important factor in the maintenance of intestinal homeostasis, live microbial supplementation (yogurt or fermented milk), have been proposed as healthy foods to control diarrheas a result of lactose malabsorption, acute viral and bacterial diarrhea, as well as for antibiotic- associated diarrhea (Heyman, 2000). Furthermore probiotics can resume the activity of colonic microflora that has been disrupted after diarrhea, or antibiotic administration (Jiang and Savaiano, 1997).
Colon cancer: According to the National Cancer Institute, cancer of the colon or rectum (colorectal) cancer, is the fourth most common cancer in men and women in United States (NIC, 2002). Fermented dairy products, that contain probiotics in yogurt, have shown an inhibitory effect on colon cancer. Heterocyclic Aromatic Amines (HAA) that are produced during the cooking of food with high creatine, free amino acid and sugar content, are known as an initiating agent of colon cancer in animals and human (Tavan et al., 2002). Tavan et al. (2002) studied the protective effect of probiotics in the fermented milk, on male F344 rats, which were used as a model of HAA- induced colon carcinogenesis. Different probiotics dairy sources that are able to decrease the incidence of colonic crypts in rats are as follows: 66% inhibition with milk- Supplement diet, 96% inhibition with Bifidobacterium animalis fermented milk-supplement diet and 93% inhibition with Streptococcus thermophilus fermented milk-supplement diet. Decrease in HAA metabolism, fecal mutagenicity and colon DNA lesions were observed. This effect being pronounced in case of milk fermented by probiotics against 1,2-Dimethylhydrazine (DMH)-induced colon carcino-genesis. Oral treatment with Lactobacillus bulgaricus against DMH-induced DNA damage in the colon in vivo, whereas S. thermophilus were not effective. However, in vitro, both strains prevented DNA damage of N-methyl-N-nitro-N-nitro-soguanidine (MNNG) carconigen, in isolated primary rat colon cells (Wollowski et al., 1999).
Inflammatory bowel disease: Inflammatory Bowel Disease (IBD) refers to a certain chronic immune- mediated condition that is characterized by acute intestinal inflammation. These chronic diseases include (ulcerative colitis, Crohn disease and pouchitis), which result from abnormal host response to some member of the intestinal flora, or from a detective mucosal barrier (Podolsky, 2002). Normally, a healthy mucosal barrier provides a first defense line against pathogens. Proportions of different intestinal microflora are altered in patients with (IBD). Colonic biopsy specimens have shown lower concentration of Lactobacillus and lower fecal concentration of both Lactobacillus and Bifidobacterium species in patient samples. Such reduction in the intestinal microflora, as well as for TNF-α pro-inflammatory cytokine, will increase the opportunity for colonization of pathogens (Borruel et al., 2002). Abnormal activation of mucosal T- lymphocytes against enteric bacteria is the key event triggering intestinal inflammation. Lactobacillus casei is able to reduce the number of activated T-lymphocytes in the lamina propria of Crohn disease, which may restore the immune homeostasis (Carol et al., 2006).
Products of probiotic and prebiotic: Probiotics are viable microbial dietary supplement, that beneficially affect the host health. Now a day probiotics are widely used to prepare fermented dairy products, such as yogurt, kefir, cheese and ice cream, or other freeze-dried cultures. In future they could be used in the fermented vegetable and meats (Roberfroid, 2000). The most functional foods that classified as prebiotics are the inulin-type fructans, which include the native hydrolyzed inulin or oligofructose and the synthetic fructooligo- saccharides (Roberfroid and Delzenne, 1998). One of the plant species Cichorium intybus that is used to produce the chicory inulin industrially. Chicory inulin and oligofructose are recognized as natural food ingredients, safety to be consumed (Roberfroid et al., 1998). Native inulin and oligo-fructose are present in significant amounts in the edible fruits and vegetables, like onion, garlic, banana and leeks (Van Loo et al., 1995). Inulin-type fructans are used as sugar substitutes, as fat replacers (inulin) and as a means of providing texture, stabilizing foams, or improving mouth feels in the miscellaneous products, such as fermented dairy products, desserts, jellies, ice creams, bakery products (bread, cookies and pastries), also it used in the infant formulas to enhance there flavors (Cummings and Roberfroid, 1997). List of probiotic strains used in commercial manufacture products (Table 4), consider as a functional ingredient benefit to be consumed (Yeung, et al., 1999).
Conclusion and recommendations: It's believed that consumption of yogurt including active Probiotics and other fermented dairy products provided with probiotics, enhance the function of the gastrointestinal tract. Many studies of the possible health benefits of yogurt in protecting against gut- associated disease Substantiate some of these beliefs. One of the most interesting preventive effects is the protection against IBD and colon cancer as suggested by epidemiologic evidence and animal studies and the therapeutic action of diarrhea caused by infectious pathogens, in addition to the beneficial effect of yogurt containing live and active culture on the digestion of lactose in patients who suffer from lactose intolerance. These findings are interesting and should encourage future studies to investigate the mechanisms of action through which yogurt exerts its effects and the critical components in yogurt that control such action. Further more, the safe history of prolong consumption of dairy product supplemented with probiotics Lactobacilli and Bifidobacteria, which are characterized by low pathogenity even if it's fed in high levels. Such finding able to encourage the diary manufacture companies to develop variety of food and feed products which provided with variable probiotics as a basic culture to enhance the final products.