Research Article
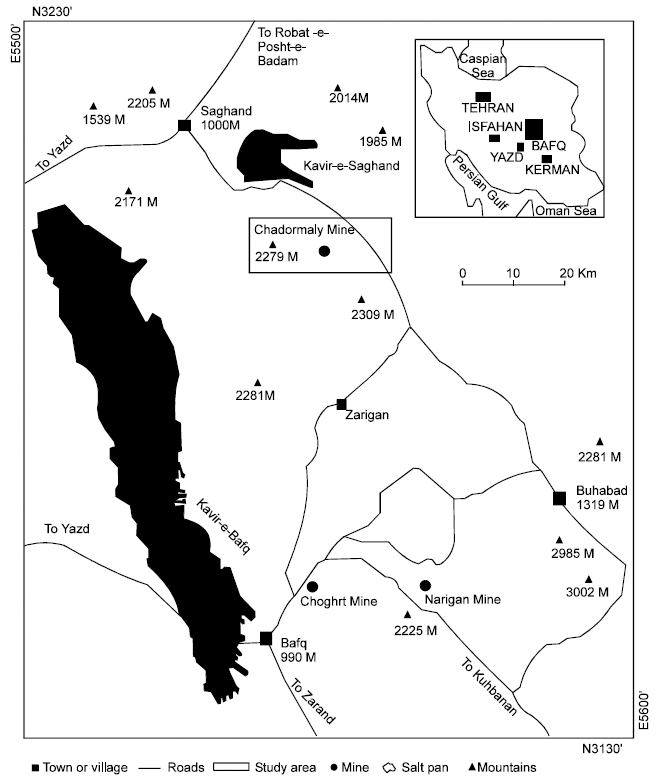
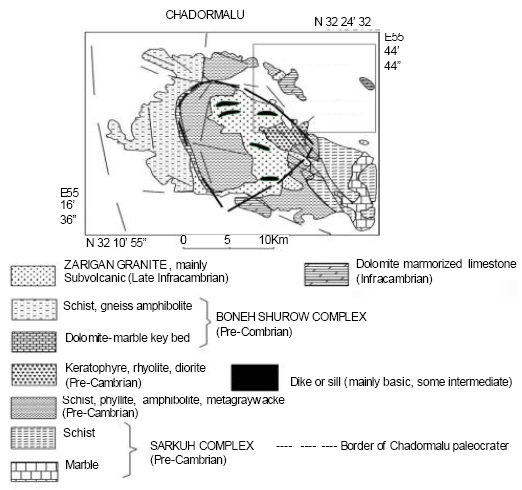
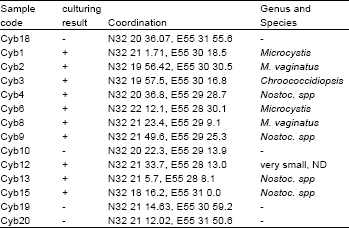
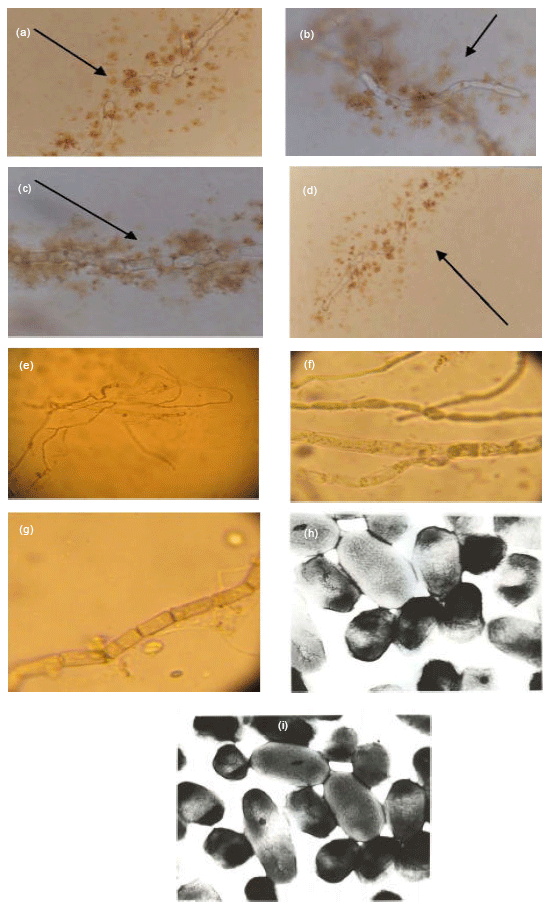
Cyanobacteria in Biological Soil Crust of Chadormalu Area, Bafq Region in Central Iran
Department of Geology, College of Sciences, Payam Noore University, Evaz, Fars province, Iran
M. Taghavi
Department of Plant Protection, College of Agriculture, Shiraz University, Shiraz, Iran
R. Rezaei
Department of Plant Protection, College of Agriculture, Shiraz University, Shiraz, Iran