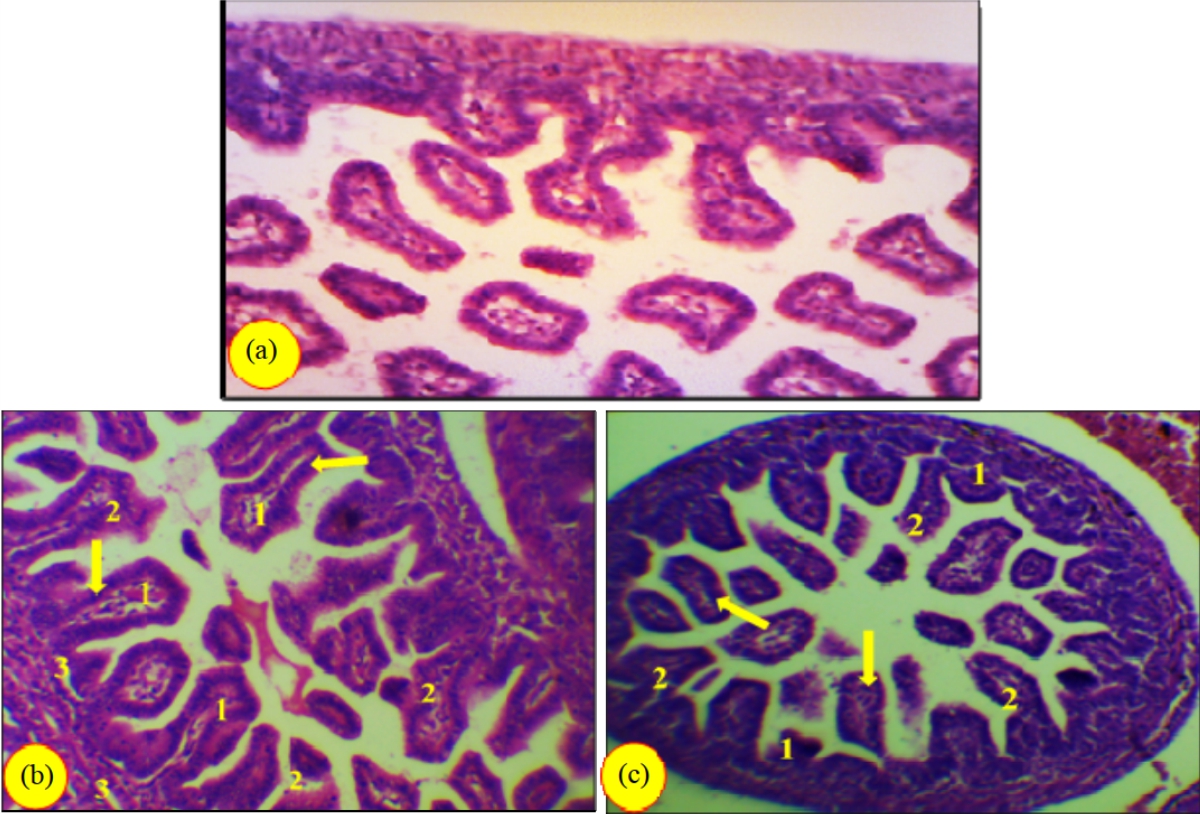
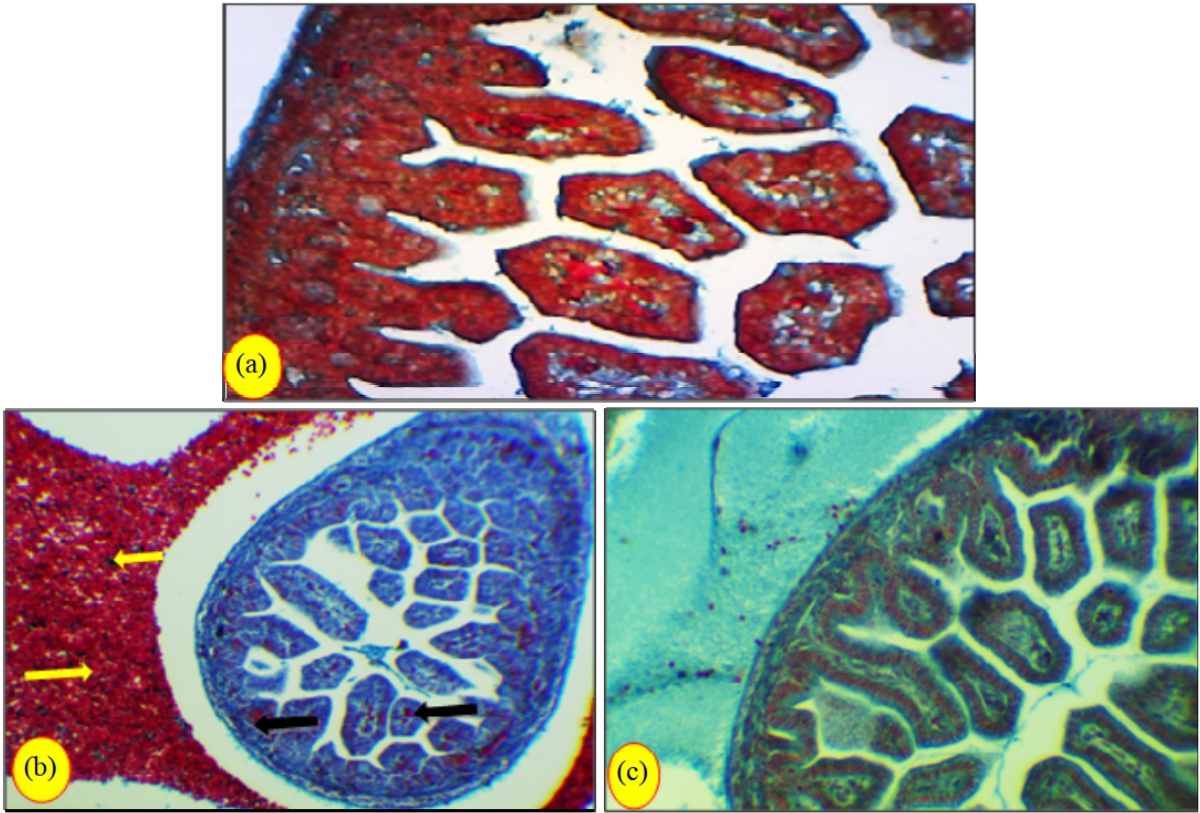
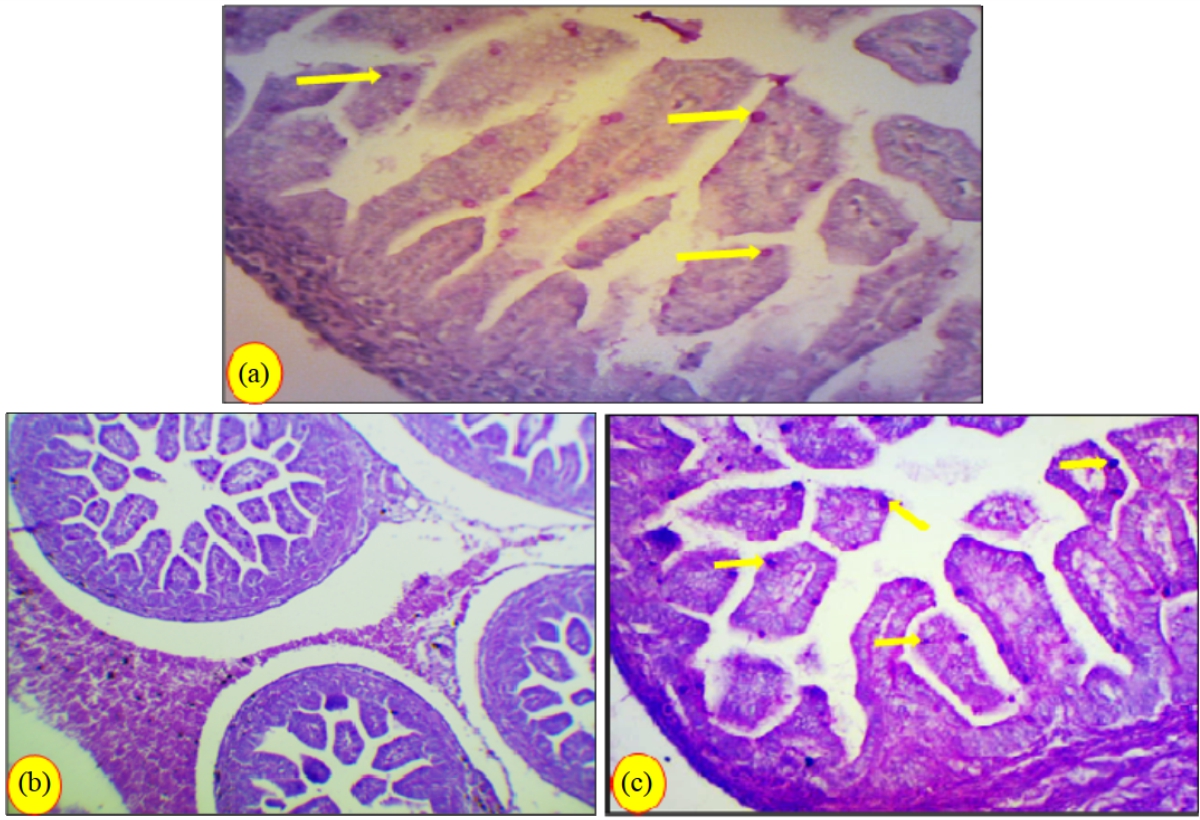
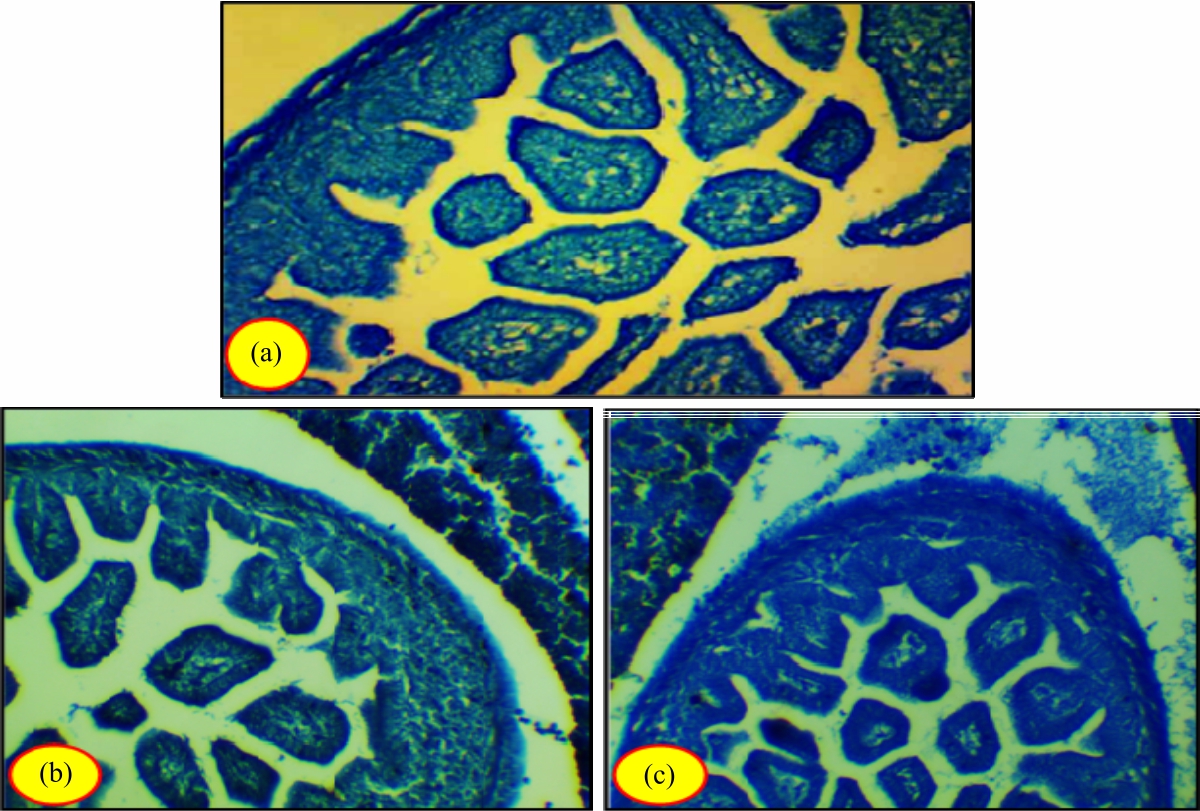
Research Article
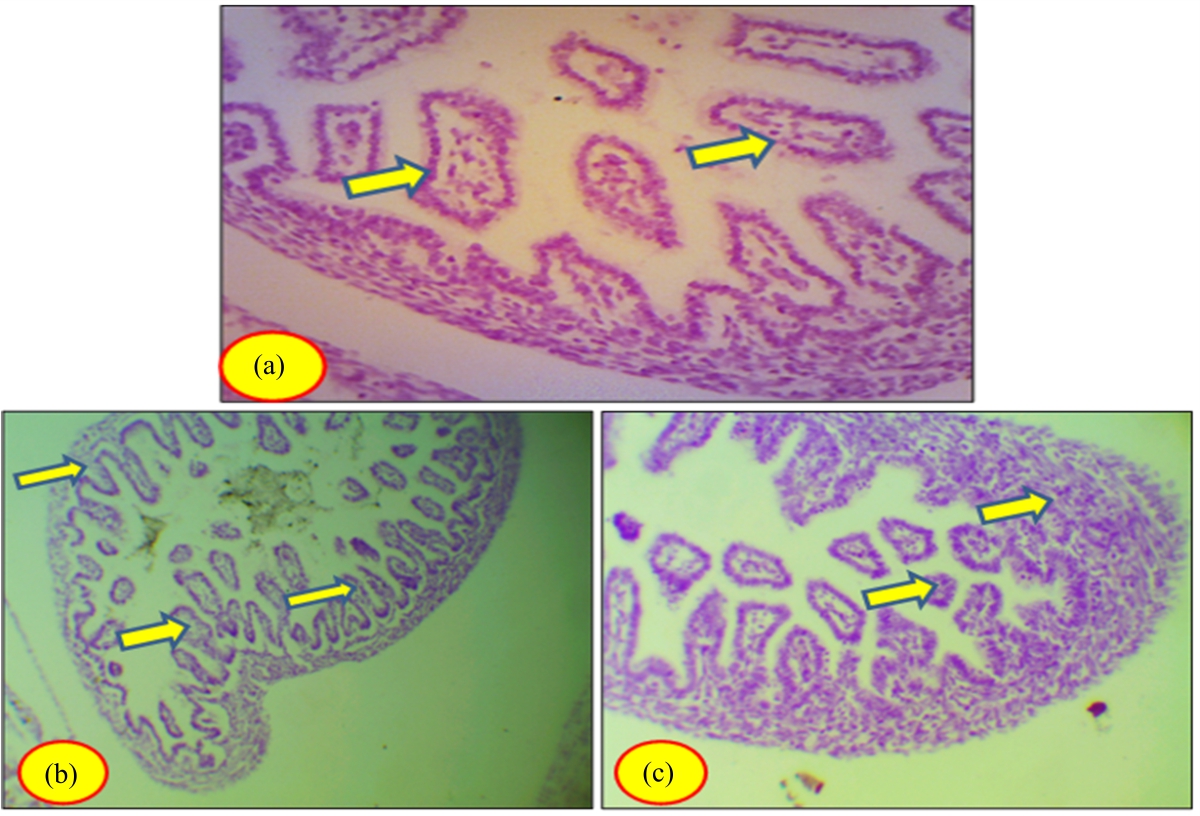
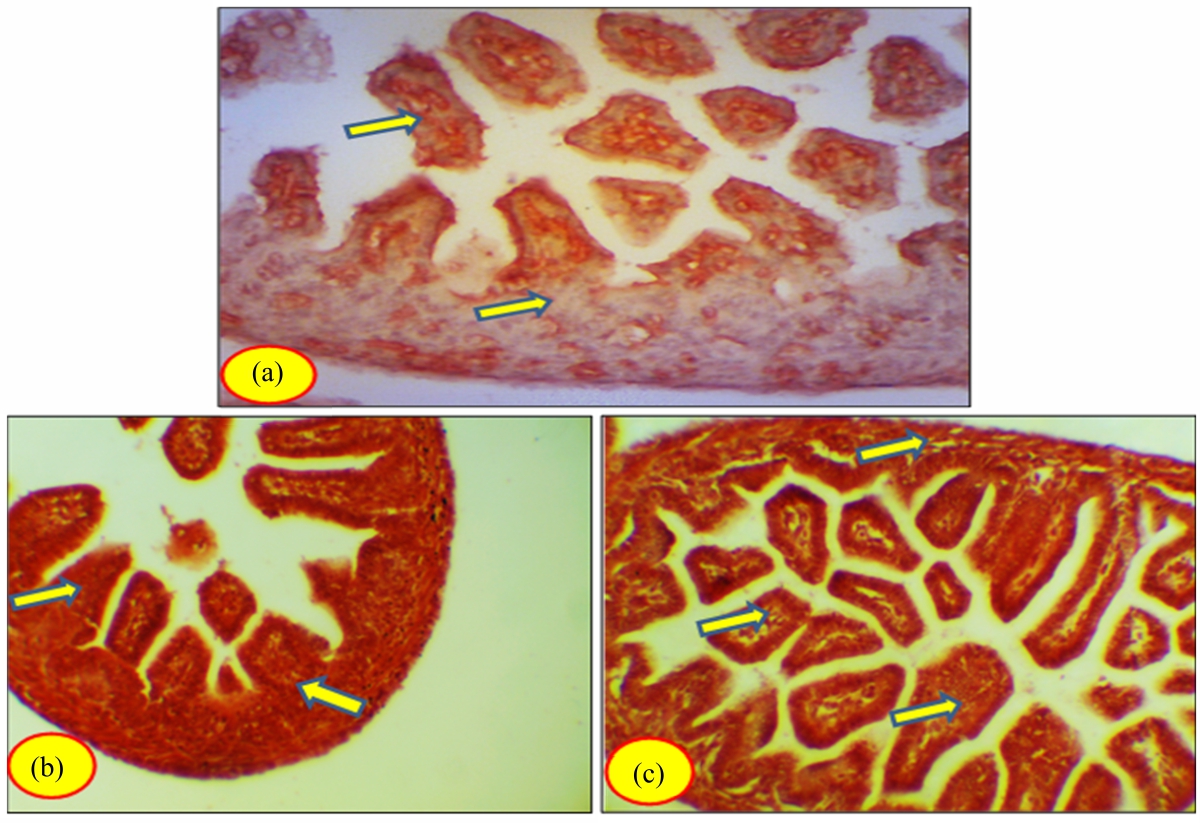
Toxicity of Somadril Compound on Fetal Ileum Tissues of Albino Rats
Biology Department, College of Science, Jouf University, P.O. Box: 2014, Sakaka, Saudi Arabia
LiveDNA: 20.22576
Mashael Alhumaidi Alotaibi
Biology Department, College of Science, Jouf University, P.O. Box: 2014, Sakaka, Saudi Arabia