Research Article
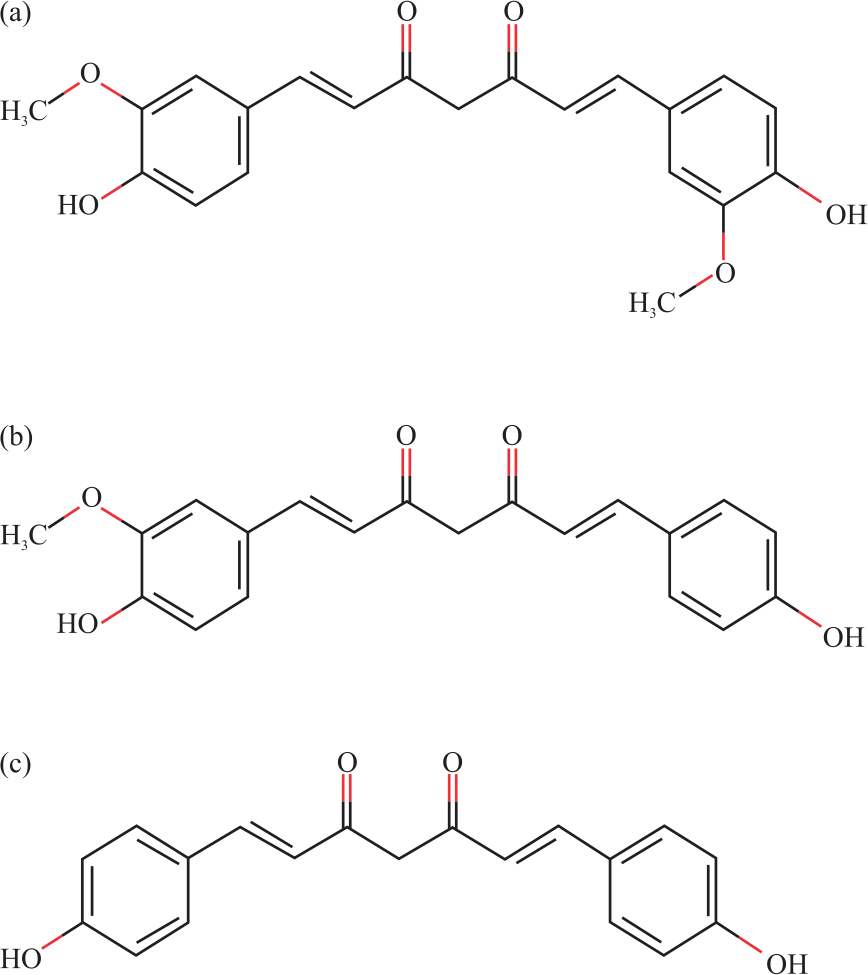
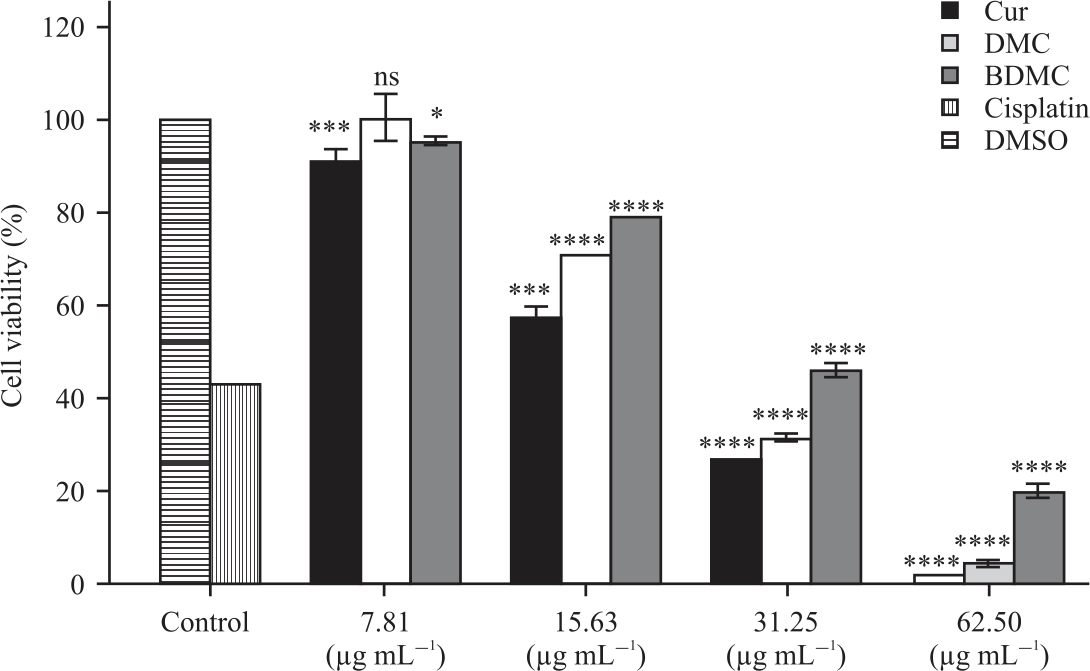
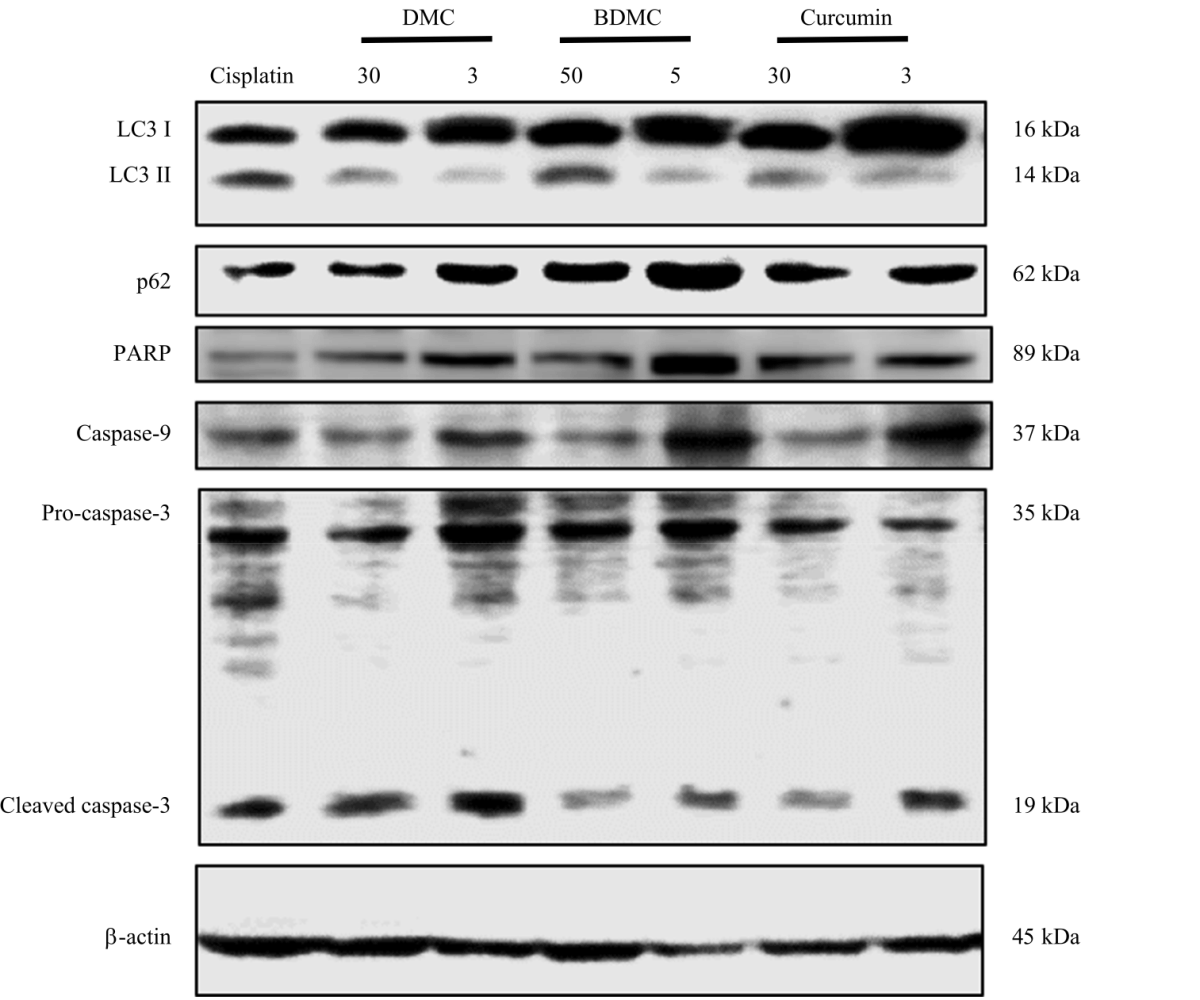
Active Compounds from Curcuma longa and Comparison of their Effectively Induced Apoptosis in MCF-7 Cell
Physiology Molecular Laboratory, Biological Activity Division, Central Laboratory, Universitas Padjadjaran, Bandung, Indonesia
LiveDNA: 62.22876
Susianti
Physiology Molecular Laboratory, Biological Activity Division, Central Laboratory, Universitas Padjadjaran, Bandung, Indonesia
Todia Pediatama
Physiology Molecular Laboratory, Biological Activity Division, Central Laboratory, Universitas Padjadjaran, Bandung, Indonesia
Nova Sylviana
Physiology Molecular Laboratory, Biological Activity Division, Central Laboratory, Universitas Padjadjaran, Bandung, Indonesia
Yuni Susanti Pratiwi
Physiology Molecular Laboratory, Biological Activity Division, Central Laboratory, Universitas Padjadjaran, Bandung, Indonesia
Hanna Goenawan
Physiology Molecular Laboratory, Biological Activity Division, Central Laboratory, Universitas Padjadjaran, Bandung, Indonesia
LiveDNA: 62.22347
Unang Supratman
Departement of Chemistry, Faculty of Mathematics and Natural Sciences, Universitas Padjadjaran, Bandung, Indonesia