Research Article
Effect of Bone Marrow Transplantation on the Fetal Skeleton of Maternally Irradiated Pregnant Rats
Department of Biology, College of Science, Jouf University, P.O. Box 2014, Sakaka, Saudi Arabia
LiveDNA: 20.22576
Exposure to radiation sources used in different appliances may be received by professionals known as radiation workers1. Radiation causes the tissue to be ionized and activated which impairs normal cell functioning2. Ionizing radiation interacts with matter and causes some modifications. These changes are known as radiation damage and cause structural changes in cells3. High doses of ionizing radiation can cause cell death due to damage in DNA strands4. The presence of injured nuclear DNA in the cells may cause cell divisions and genomic instability5. Radiation exposure thus causes mutagenic effects and organ-specific changes6.
Gamma irradiation was also found to cause embryonic loss and malformations in mice7. During the organogenesis period in pregnancy, radiation may cause malformations in some organs and possibly growth retardation, death of fetuses, mental retardation and microcephaly8. The skeletal malformations in pregnant mice were noted when exposed to a single dose of 2.0 Gy on days 2 and 15 of gestation9. Skeletal elements of mice fetuses maternally exposed to gamma rays showed very poor ossification in the system especially during the organogenesis period10. On day 3, 10 and 14 of gestation, pregnant rats had a lower oscillation in the skull bones, less oscillation in the vertebral centra and wavy ribs at a single sublethal dose level of 4 Gy7.
Exposure of gravid mice to electromagnetic field radiation indicated a temporary disruption in the ossification process of different bones, which only improves after giving birth11. The female mice exposed to 1 Gy of protons irradiation at sixteen weeks old caused the loss of trabecular bone fraction in the tibia and femur12. Bone marrow is a complex tissue comprising two hematopoietic and stromal sections. The stromal division is a complex of tissue a hematopoietic microenvironment containing a group of cells called mesenchymal stem cells (MSCs)13.
In recent years, stem cell strategies have become very popular. Bone marrow MSCs can distinguish between many mesenchymal types of cells, including cartilage, bone and fat cells14. Mesenchymal stem cells are used to heal injured tissue and other tissue damage, including damage within the cardiac system, the nervous system and lunate bone15. Transplantation of bone marrow increases the level of antioxidants and protects rats from oxidative stress16.
The current study was done to study the probable protecting effect of BMT on the skeleton of fetuses of maternally irradiated pregnant rats.
Study area: This research was done at Atomic Energy Authority, National Center for Radiation Research and Technology, Cairo, Egypt from January-March, 2013.
Irradiation: Irradiation was provided by applying Gamma-cell 40 (137 Cesium), at the Atomic Energy Authority, National Center for Radiation Research and Technology, Cairo, Egypt.
Experimental animals: Albino rats (Rattus albinus) kept in cages with body weights ranging from 120-150 gm were used. The males were separated from females until mating. Two females of estrous or proestrus periods were confined with one male. The pregnancy was assured the next morning by the presence of a plugin the vagina or the occurrence of spermatozoa in the smear of vaginal content and that day represented the 1st day of gestation.
Bone marrow transplantation: Bone marrow transplantation was of the same strain (receptors and donors). The donors were sacrificed and femur bones were gutted and both ends were disjointed. The marrow was put into saline solution under sterilized conditions then mixed and expelled from the syringe several times without a needle to prevent the cells from mechanical damage. The number of cells about 75×106±5 were inoculated 1 h after irradiation17. The bone marrow was injected intraperitoneally in the pregnant rats.
Groups of animals: The gravid rats were separated into 5 groups (six rats in each group), C set (control gravid rats), R7 set (Gravid rats exposed to one dose, two grays of γ rays on day 7 of pregnancy), Group R7+BM (Pregnant rats exposed to one dose, two grays of γ rays on the 7th day of gestation then received bone marrow transplantation (75×106±5 cells) via intraperitoneal dose 1 h after radiation, R14 group (Pregnant rats exposed to one dose, two grays of γ rays on the 14th day of pregnancy), R14+BM group (Pregnant rats exposed to one dose, two grays of γ rays on the 14th day of gestation and then received bone marrow transplantation (75×106±5 cells) via intraperitoneal dose 1 h after radiation. All gravid rats were sacrificed on the 20th day of pregnancy.
Skeletal studies: Alizarin red stain was used for evaluating the endoskeletal system of fetuses. The endoskeletons were prepared according to the method of Chahoud and Paumgartten18. The skeletal systems were then examined and photographed. This study focused on evaluating the following regions the skull region, upper and lower jaw, occipital region, sacral and caudal region, forelimb and hind limb.
The normal skull of a rat is consists of the premaxilla, maxilla, nasal, frontal, parietal, intraparietal, squamosal, jugal, exoccipital, supraoccipital, basioccipital and tympanicum. (Fig. 1a). Fetuses of the R7 group showed a wide suture between the two parietal bones (Fig. 1b) but the treatment of this group with bone marrow 1 h post-irradiation reconstructed the suture between the dermal bones with dense ossification centers in them (Fig. 1c). Less ossification in the frontal and parietal bones was observed in fetuses of group R14 (Fig. 1d). The fetuses of group R14+BM showed conspicuous improvement in ossification centers with a wide suture in the midline of the cranium (Fig. 1e).
The Fig. 2a showed normal ossification in the nasal, premaxilla, maxilla and zygomatic arch. Shortness in the premaxilla bone was observed in the fetuses of the R7 group (Fig. 2b).
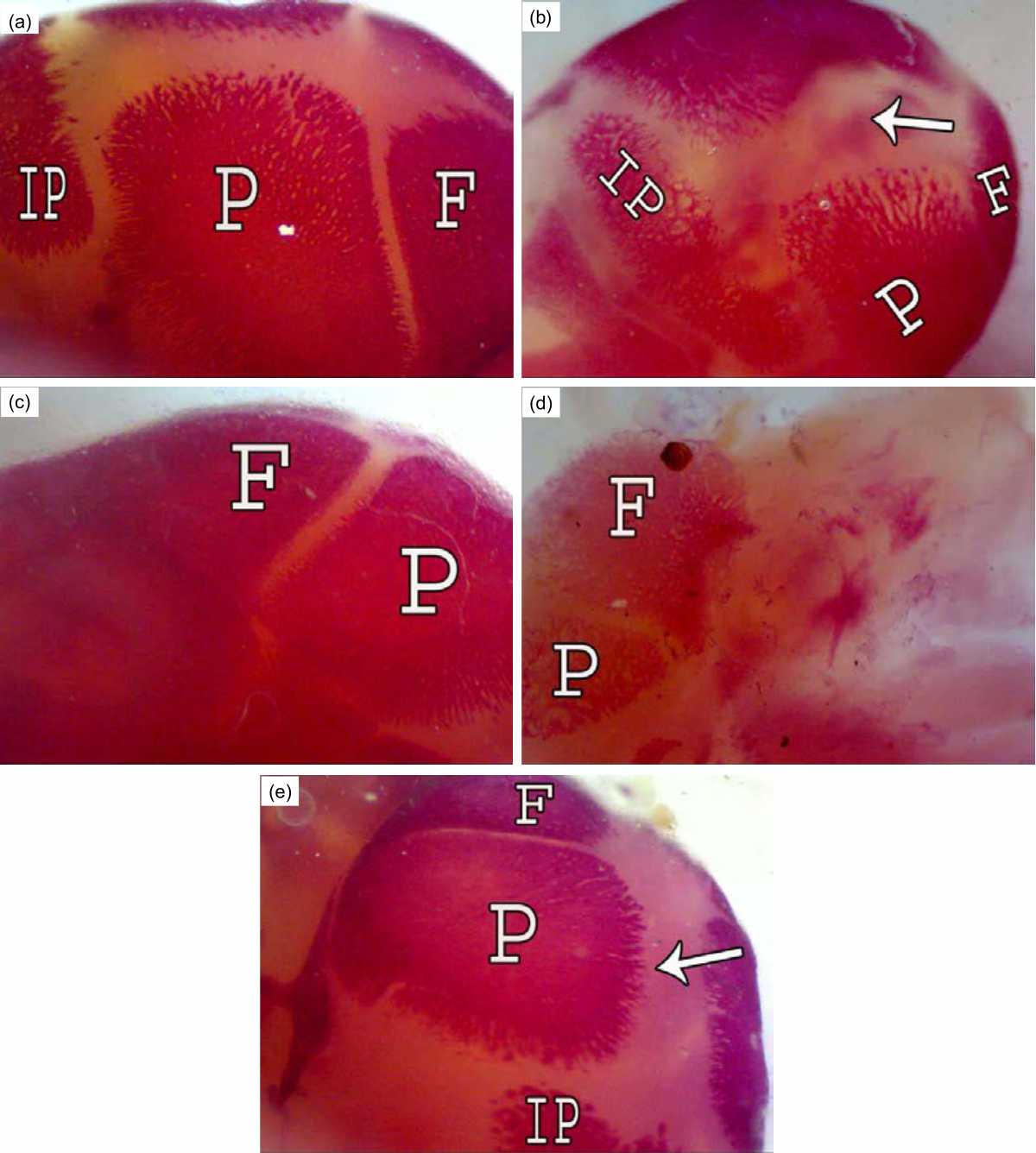 |
| Fig. 1(a-e): | Cranium of rat fetuses after 20 days of gestation A (control group): Normal ossification of the dermal bones frontal (F), parietal (P) and interparietal (IP), B (R7 group): A large suture between two parietal bones (↑), C (R7+BM group): A dense ossification of parietal and frontal bones, D (R14 group): Less ossification in the frontal and parietal bones and all the bones with high porosity, E (R14+BM): Conspicuous improvement in ossification of the dermal bones with large suture in the mild line of the cranium (↑) |
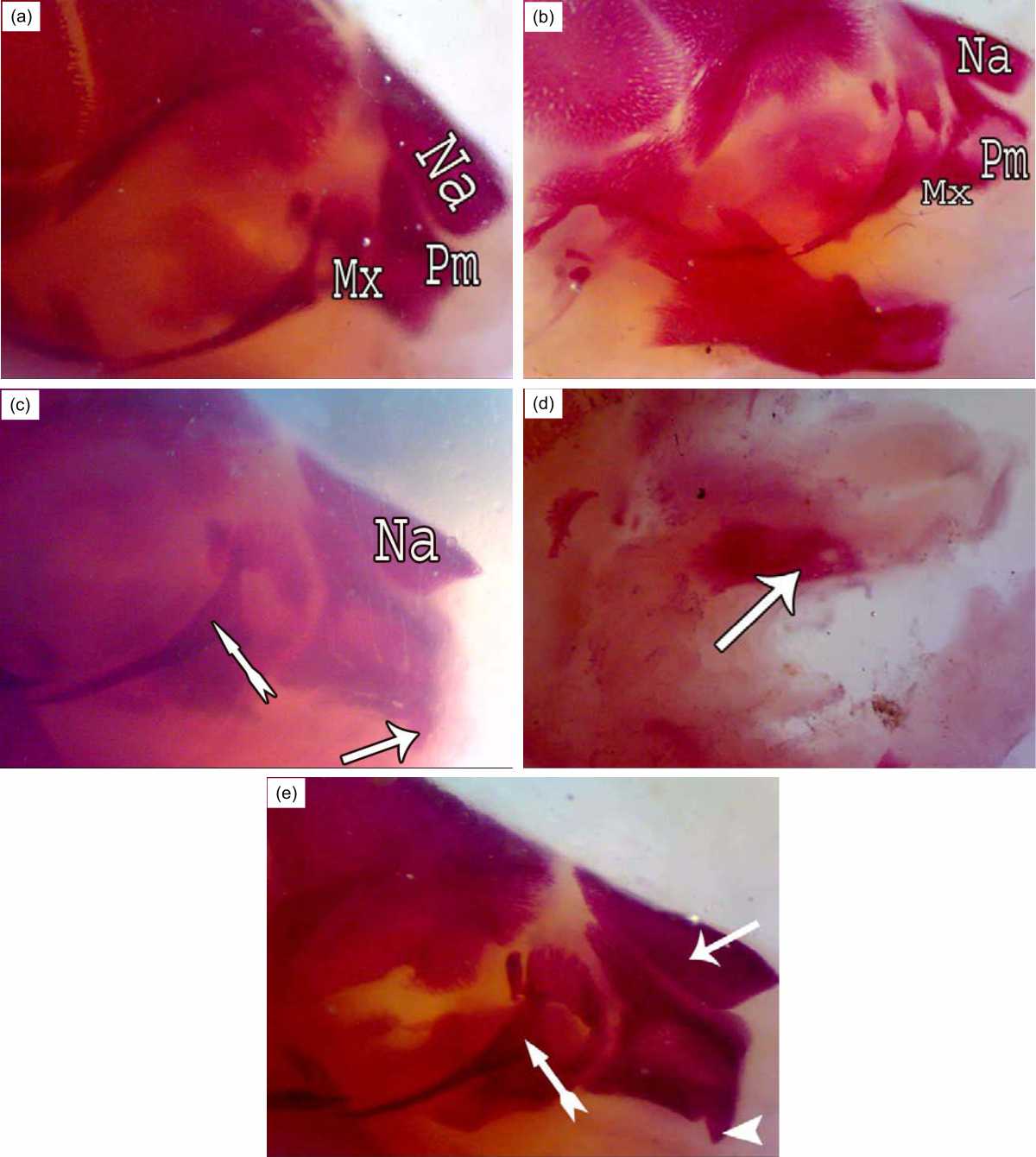 |
| Fig. 2(a-e): | Upper jaw of rat fetuses after 20 days of gestation A (control group): conspicuous ossification in the Premaxilla (Pm), Maxilla (Mx), B (R7 group): Shortness in the premaxilla bone (Pm), C (R7+BM group): Long and thin apex of nasal bone (Na) like rostrum, presence of incisor (↑) and thick zygomatic process of maxilla (»); D (R14 group): Part of maxilla was ossified only (↑); E (R14+BM): Elongated nasal bones (↑) ,presence of incisor (▴) and thick zygomatic process of maxilla (») |
Post-irradiation, bone marrow transplantation in this group showed the nasal summit resembling the beak and the increase in incisor development and the thick zygomatic process of the maxilla (Fig. 2c). Fetuses in the group R14 showed only an ossified part of the maxilla bone (Fig. 2d), while fetuses of the R14+BM group showed elongated beak-like nasal summit bone and evolution in the incisor growth (Fig. 2e).
Conspicuous ossification of the mandible bone was observed in the control fetuses (Fig. 3a). Irradiation on the 7th day of gestation caused shortness in this bone (Fig. 3b). Fetuses of the R7+BM group showed elongated mandible bone and evolution in the incisor growth (Fig. 3c). Fetuses of group R14 displayed no ossification centers in the mandible bone (Fig. 3d) but the bone marrow transplantation after exposure to gamma rays on the 14th of gestation accelerated the development of the mandible and incisor (Fig. 3e).
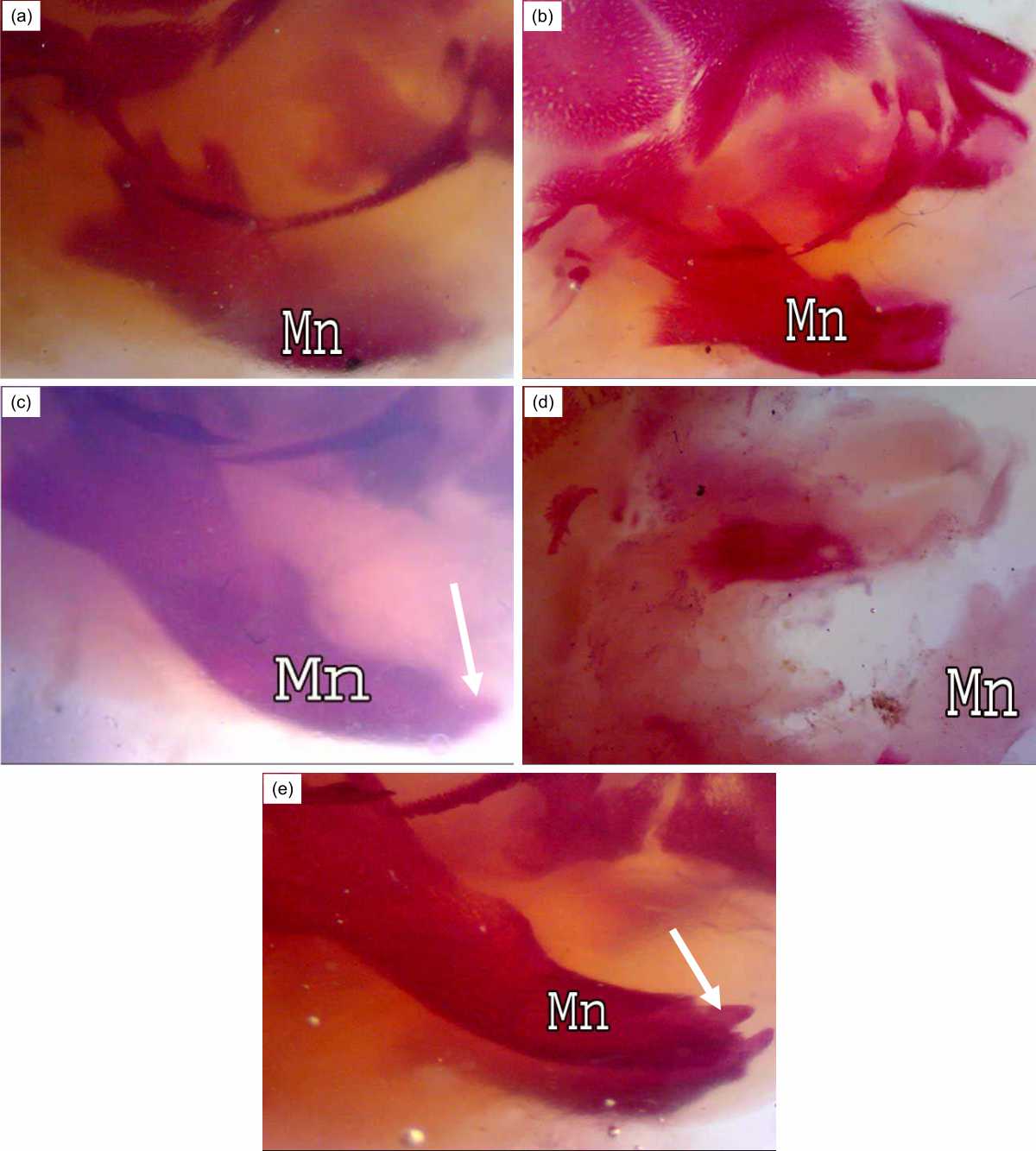 |
| Fig. 3(a-e): | Lower jaw of rat fetuses after 20 days of gestation A (control group): Conspicuous ossification of mandible bone (Mn), B (R7 group): Shortness in the mandible bone, C (R7+BM group): Elongated mandible bone, D (R14 group): Unossified mandible bone, E (R14+BM): Good ossification in the mandible bone, Note, bone marrow transplantation accelerate the development of mandible and incisor formation (↑) in groups R7+BM and R14+BM |
The Fig. 4 showed the occipital region in the normal and treated groups. Fetuses of the control group displayed conspicuous ossification in the supraoccipital, exoccipital and atlas bones (Fig. 4a). The growth rate of the supraoccipital, exoccipital and atlas bones was reduced in the fetuses of the R7 group (Fig. 4b). Bone marrow transplantation one-hour post-irradiation caused thickness in the supraoccipital bone with improvement in the wing sides (Fig. 4c). Fetuses of group R14 showed absence of ossification centers in the supraoccipital, exoccipital and atlas bones (Fig. 4d), while normal ossification centers were observed in the occipital region of the fetuses of the R14+BM group (Fig. 4e).
The Fig. 5 showed the sacral and caudal regions of rat fetuses after 20 days of gestation, Fig. 5a normal control.
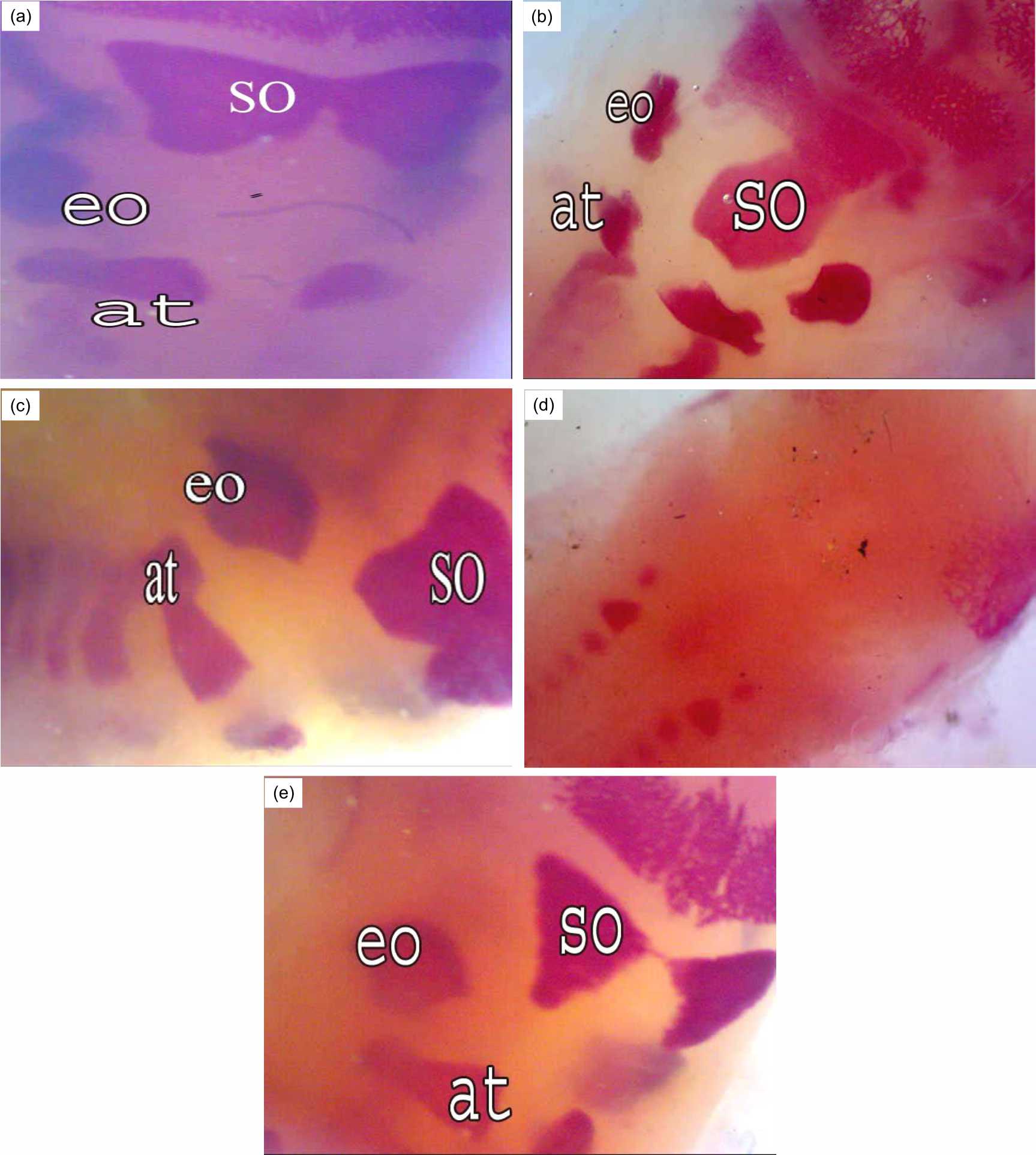 |
| Fig. 4(a-e): | Occipital region of rat fetuses after 20 days of gestation A (control group), conspicuous ossification in the supra occipital (so), ex occipital (eo) and atlas (at), B (R7 group): Reduction in the size of supra occipital, ex occipital and atlas bones, C (R7+BM group): improvement in ossification of occipital bones (so, eo and at), D (R14 group): Absence of ossification centers in the supra occipital, ex occipital and atlas, E (R14+BM): Normal ossification in the occipital bones |
Fetuses of group R7 showed five sacral vertebrae were ossified but the caudal vertebrae ossification varies and only one caudal piece was ossified (Fig. 5b). Fetuses of R7+BM group like control (Fig. 5c). The sacral and the caudal vertebrae are non-ossified in the R14 group (Fig. 5d). Good ossification as in the control group was found in the R14+BM group (Fig. 5e).
The pectoral girdle (scapula, supra scapula and clavicle), in the fetuses at 20 days of gestation, showed complete ossification in the normal and treated fetuses of different sizes. Bones of the forelimb (hummers, radius, ulna, 5 metacarpals and phalanges) were detected in the normal fetuses (Fig. 6a). Incomplete ossification in the metacarpal bones (3 only) was observed in the fetuses of the R7 group (Fig. 6b). Bone marrow transplantation post-irradiation on the 7th day of pregnancy caused an increase of the number of metacarpals (4), phalanges and nails (Fig. 6c).
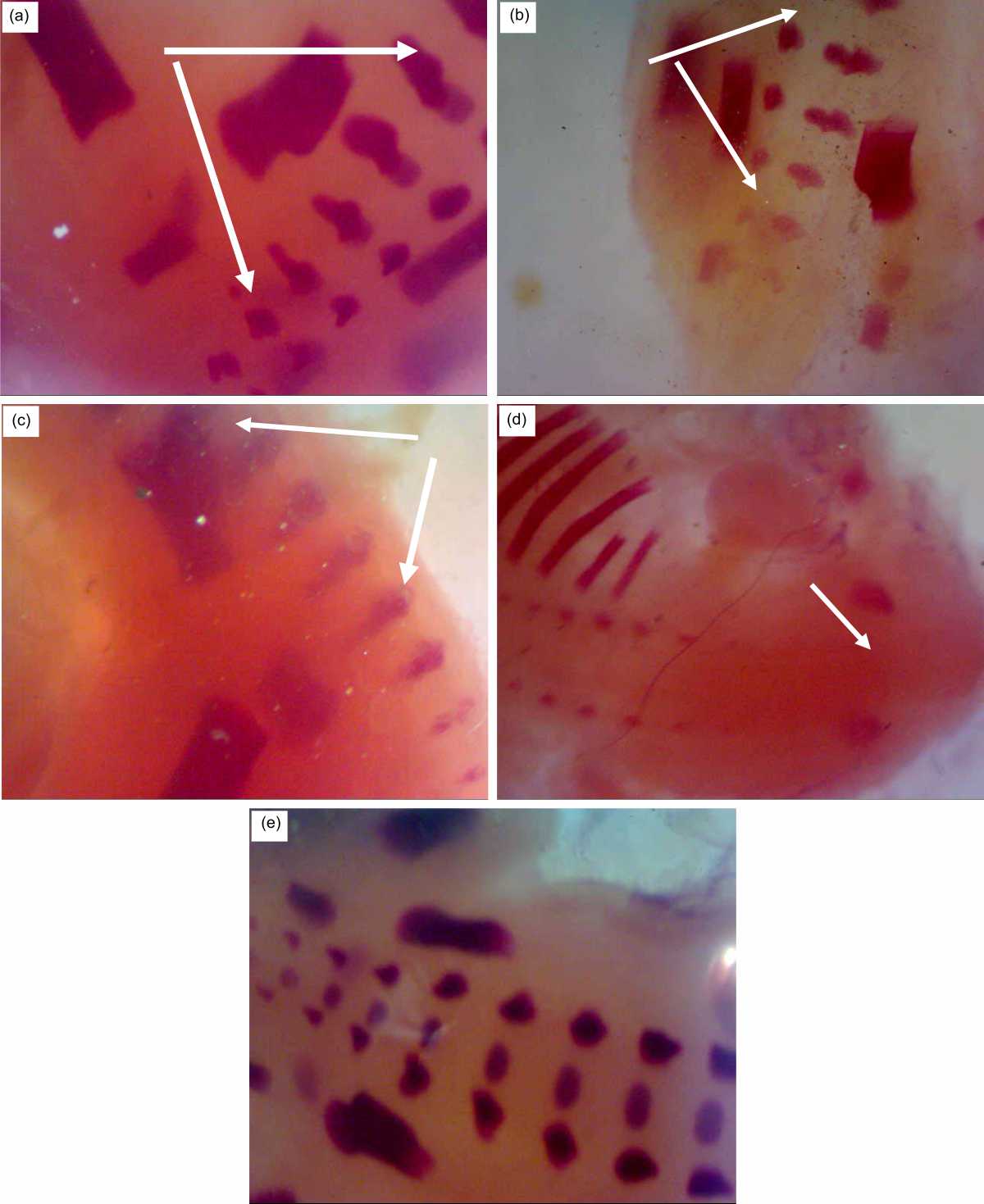 |
| Fig. 5(a-e): | Sacral and caudal regions of rat fetuses after 20 days of gestation A (control group): Five sacral vertebrae are ossified (↑), but the caudal vertebrae are variable B (R7 group), only one caudal piece is ossified, C (R7+BM group) like control, D (R14 group), the sacral and the caudal vertebrae are non-ossified (↑), E (R14+BM), good ossification as in the control group |
Fetuses maternally irradiated on the 14th of gestation displayed incomplete ossification in the pectoral girdle as well as in the humerus, radius and ulna with the absence of ossification centers in the metacarpals and phalanges (Fig. 6d). Normal ossification centers were observed in the pectoral girdle and forelimb after bone marrow transplantation in this group (Fig. 6e).
As Fig. 7a showing normal bones of the hind limb (femur, tibia and fibula) in the control and maternally treated fetuses.
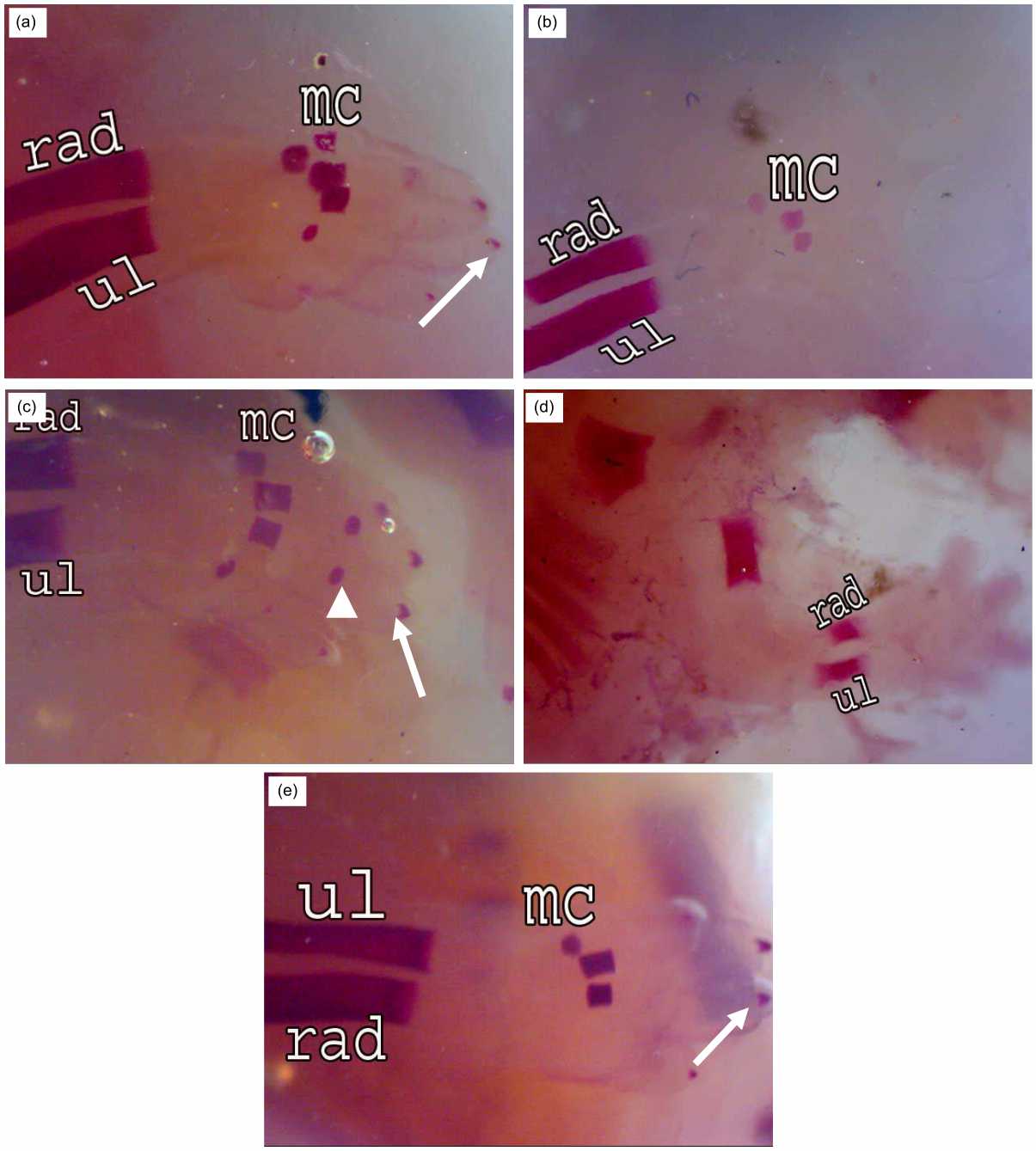 |
| Fig. 6(a-e): | Fore limb of rat fetuses after 20 days of gestation A (control group): Conspicuous ossification in the Radius (rad), Ulna (ul) and 5 Metacarpals (mc), weak ossification in nails (↑), B (R7 group): Incomplete ossification in metacarpal bones (3), C (R7+BM group): Improvement in the number of metacarpals (4), phalanges (▴) and nails (↑), D (R14 group): Incomplete ossification in the radius, ulna and absence of metacarpals, E (R14+BM): Good ossification in metacarpals (3) and nails (↑) |
The ossified pieces of metatarsals were 4 in the normal fetuses and the phalanges were non-ossified. Fewer ossification centers were observed in the metatarsal bones (3) of the fetuses whose mother was exposed to gamma irradiation on the 7th day of pregnancy (Fig. 7b). Bone marrow transplantation post-irradiation on the 7th day of pregnancy indicated progress in ossification centers of the metatarsal bones (4) (Fig. 7c).
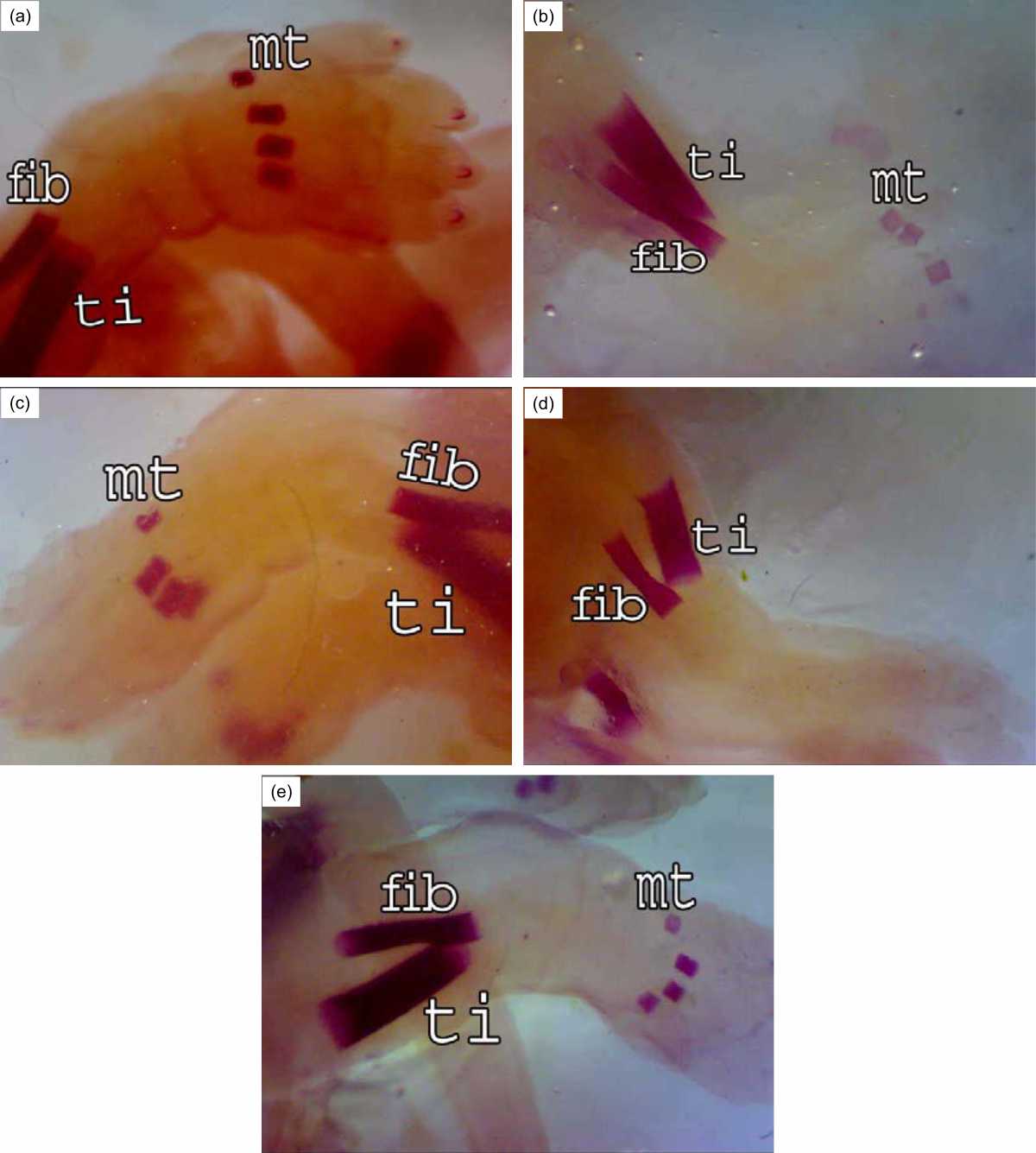 |
| Fig. 7(a-e): | Hind limb of rat fetuses after 20 days of gestation A (control group), good ossification in the Fibula (fib), Tibia (ti) and metatarsus(mt) (4), B (R7 group), less ossification in the metatarsus (3), C (R7+BM group), Improvement in the ossification centers of the metatarsus (4), D (R14 group), absence of ossification centers in the metatarsus, E (R14+BM), the ossification centers of the shank and foot are like normal |
Fetuses of group R14 showed the absence of ossification centers in the metatarsal bones (Fig. 7d) but normal ossification centers were observed in fibula, tibia and metatarsal bones (4) after bone marrow transplantation in this group (Fig. 7e)
The results of the present study indicated disturbances in the ossification process of the skull bones, upper and lower jaws, occipital bones, as well as the absence of some ossification centers metacarpal bones, metatarsal bones in the fetuses of pregnant rats irradiated on the 7th or 14th of pregnancy. These outcomes agree with those of Fragopoulou et al.11. Less ossification was noted in the various bones of fetuses of gravid rats irradiated with 4Gy of γ-rays on the 3rd, 10th and 14th days of pregnancy7.
Gamma radiation is shown to damage bone tissue through a free and radical effect on collagen19. It has been shown that therapeutic doses of radiation have deleterious effects on the health of bone tissue, sometimes causing spontaneous fractures and osteoradionecrosis20.
As for pregnant mice exposed to radiation from days 7-10 of pregnancy, it is believed that the development of embryos is vulnerable to the outside environment and since limb development is important for the quality of life the observed increased rates of polydactyly fetuses is troubling21.
Skeletal elements of mice fetuses maternally exposed to gamma rays showed very poor ossification in the system after exposure during organogenesis followed by fetal and pre-implantation periods10.
A decrease in the bone calcium content of rats exposed to 5 or 10 Gy of gamma rays led to increased skeletal malformation22. The drop in calcium in the bone may be due to the demineralization of the bone after irradiation23.
The skeletal elements play roles in joined craniofacial morphogenesis. Because of the highly complex nature of head development, craniofacial dysplasias alone account for many dimorphic features. Any abnormality or delay in the development of embryonic cranial constitutes skeletal deviation abnormality24. Furthermore, ionizing radiation has been implicated in osteoporotic phenotypes, bone loss and fracture risk25.
Multipotent cells which are called human mesenchymal stem cells prove valuable for the treatment of degenerative diseases. MSCs in many tissues and organs can be found between differentiated cells but, unfortunately, a phenotypical similarity impedes robust cells and discrimination from different tissue harvest26.
The first experimental use of Bone Marrow Transplantation was made for people exposed to radiation after a nuclear accident27. Transplanted bone marrow successfully replaced marrow damaged by irradiation. Transplantation of the bone marrow is not a surgical procedure but a painless infusion equivalent to a blood transfusion in the hospital.
In the present study, BMT post-irradiation indicated a noticeable improvement in the bones of the skull, upper and lower jaws, fore limb and hind limb.
MSCs are alleged to be able to distinguish into cardiomyocytes, myoblasts and neurons. Formation of cells of the non-mesodermal source may be an outcome of a phenomenon called “stem cell plasticity”, a process of transdifferentiation that is not limited to forming differentiated cell tissue types in which Organ-specific stem cells are located28.
Osteogenesis needs MSCs to be incubated with ascorbic acid phosphate, β-glycerol-phosphate, fetal bovine serum and dexamethasone. MSCs should reveal osteoplastic morphology together with a high appearance of alkaline phosphatase and calcium deposit29.
The hematopoietic stem cell pool is dispersed through the places of hematopoietic cell reconstruction in a great number of bone marrow units dispersed throughout the skeleton. Bone marrow sites that are haematopoietically active function as an organ system through the stem cell migration streams that link all sites via blood and guarantee a local stem cell level which ensures a balance between cell growth and cell removal is maintained30.
Bone marrow energizing made more chondrogenesis in Trochlea (TR) and Medial Femoral Condyle (MFC) blot in adult rabbits, with further chondrocytes and larger chondrogenic foci seeming in MFC and TR versus on the 21st day post-operation31.
Bone marrow transplanted cells to osteogenesis imperfecta patients and showed that about 2% of the osteoblasts in the recipient's bone marrow came from the donor. MSCs can become normal osteoblasts and contribute to the accelerated growth of bones and reduced rates of fracture32. Stem cell transplantation can treat osteogenesis imperfecta. Due to the irregular bone form, bone growth was sluggish, bone fractures were common and bone defects occurred33.
The application of stem cells populations to bone engineering strategies has the potential to deeply influence regenerative medicine34. Stem cells from bone marrow correct and restore a proper microenvironment to improve transplantation procedures and general disease outcomes35. The study recommends that the pregnant must not be exposed to radiation during pregnancy, so that may causes defects in the skeleton of fetuses.
This study revealed the presence of instabilities in the process of ossification in the skull bones, upper and lower jaws, occipital bones of fetal rats maternally irradiated pregnant rats, as well as the absence of some ossification centers in the frontal and parietal bones and all the bones with high porosity, conspicuous ossification in the radius and ulna, absence of ossification centers in the metatarsus. Bone marrow transplantation post-irradiation indicated a noticeable improvement in the bones of the skull, upper and lower jaws, forelimb and hind limb.
This study discovers the possible protective role of bone marrow transplantation in decreasing the gamma radiation hazards in the skeleton of fetal rat maternally irradiated. This study will help the researcher to avoid radiation during pregnancy periods and the importance of bone marrow transplantation in recovering these disturbances. Thus, a new theory has to introduce to injury of radiation on the skeleton of fetal rats.
Author wish to thank Dr. Omaima Ashry, National Center for Radiation Research and Technology, Cairo, Egypt for the qualified support to the fieldwork. The author is deeply grateful to Dr. Nehal Abo El Naga and Dr. Fatma Eid for their valuable support in this study.