Research Article
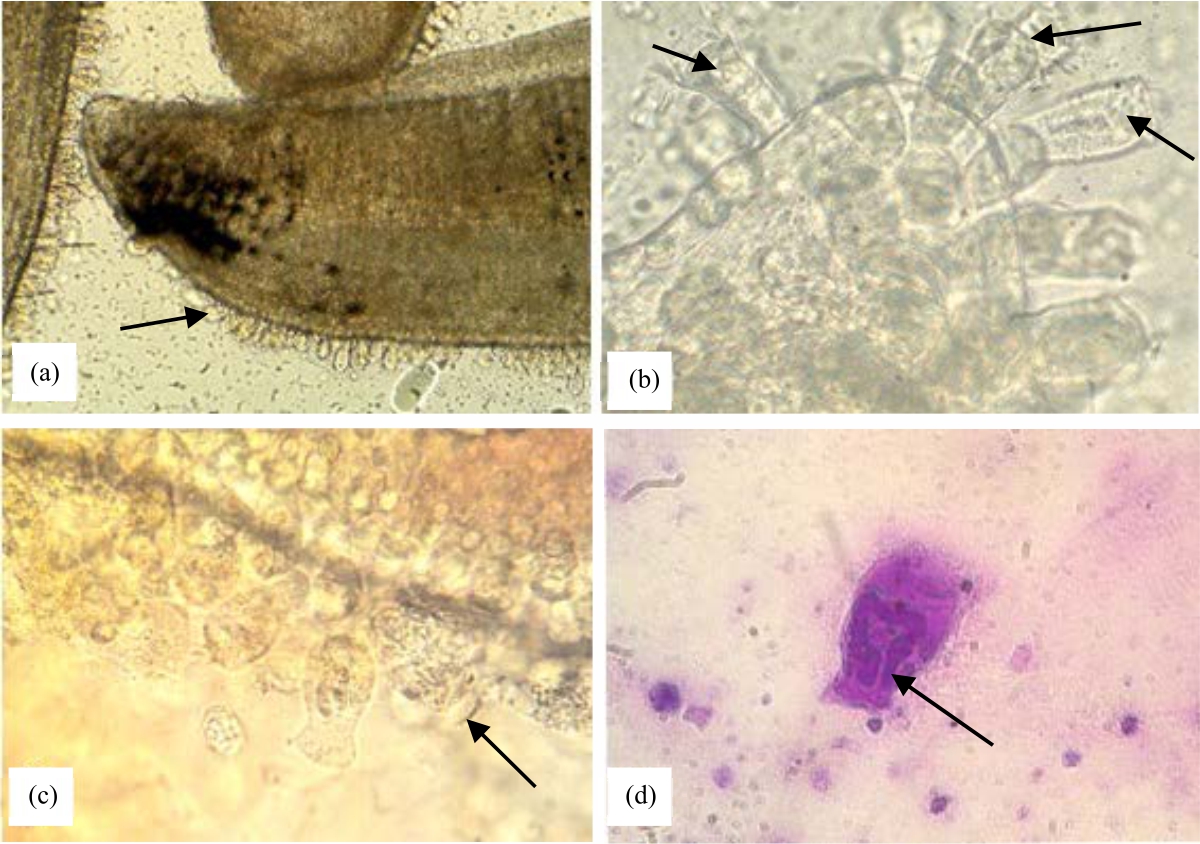
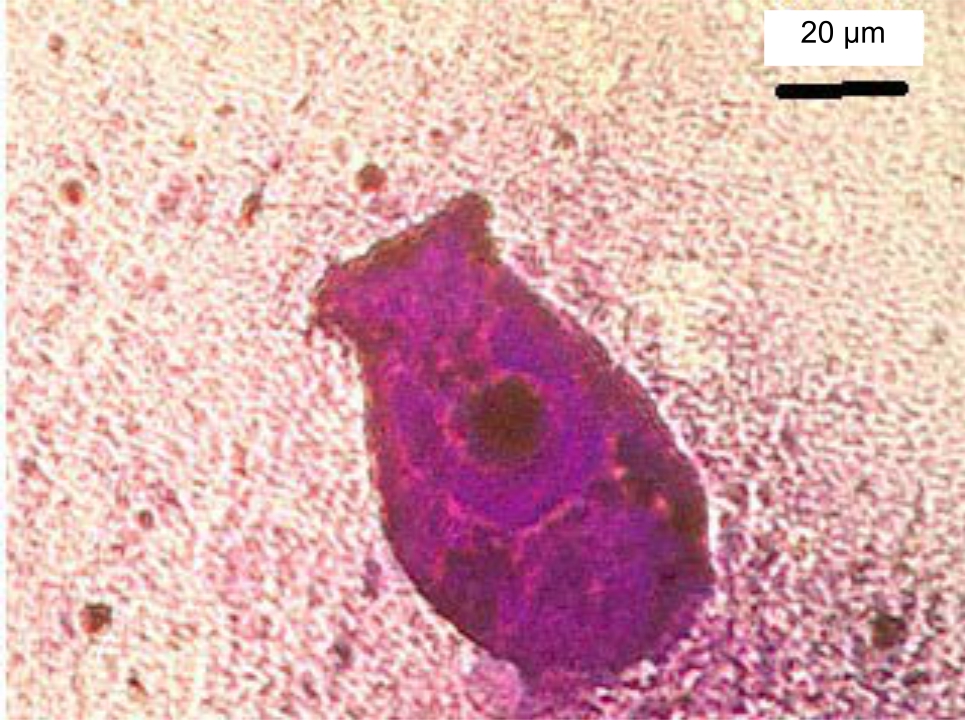
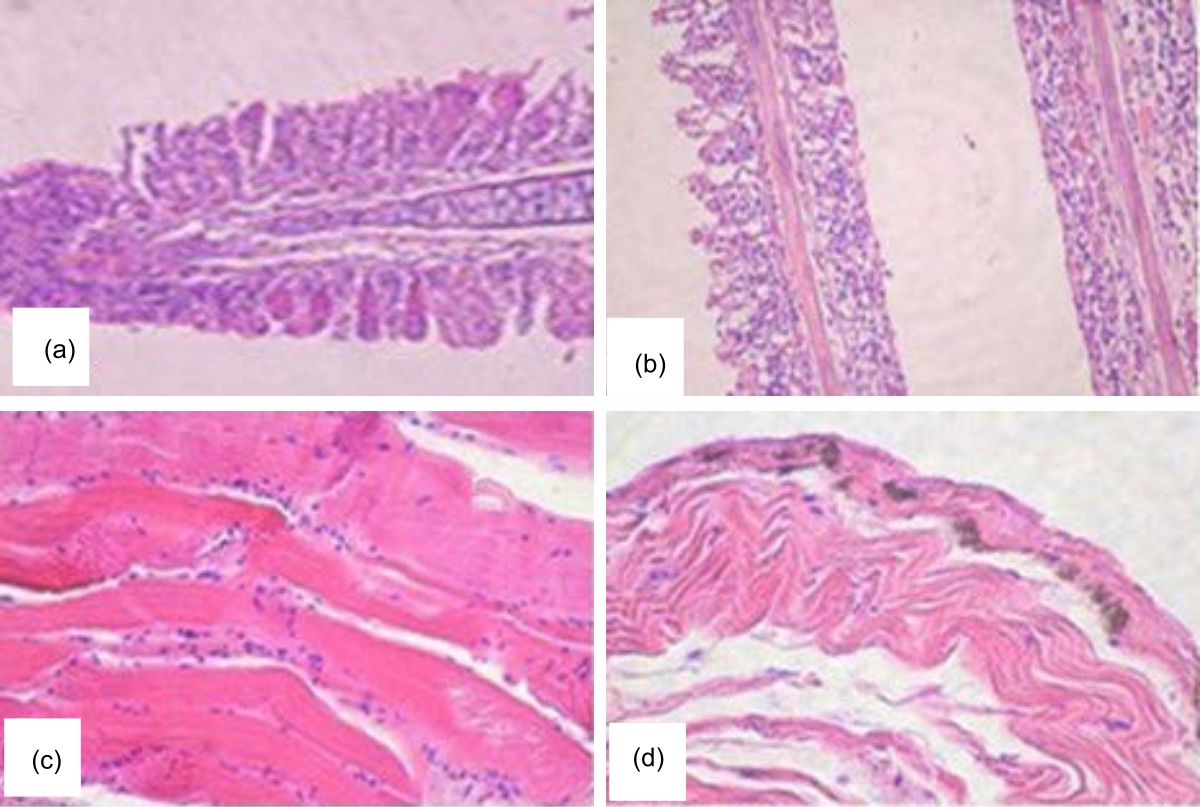
Control of Riboscyphidia sp. (Ciliate) Infection in Asian Sea Bass (Lates calcarifer), Cultivated in the Red Sea
Department of Hydrobiology, National Research Centre, Dokki, Giza, Egypt
LiveDNA: 20.21228
Abeer E. Mahmoud
Department of fish diseases, Animal Health Research Institute, Assuit Laboratory, Agriculture Research Center, Dokki, Egypt
Amany M. Kenawy
Department of Hydrobiology, National Research Centre, Dokki, Giza, Egypt