Research Article
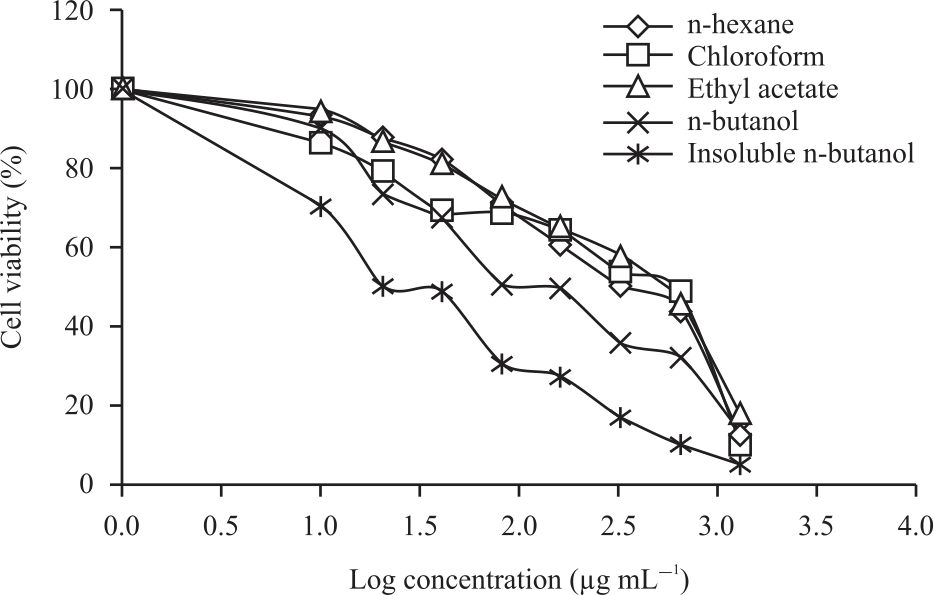
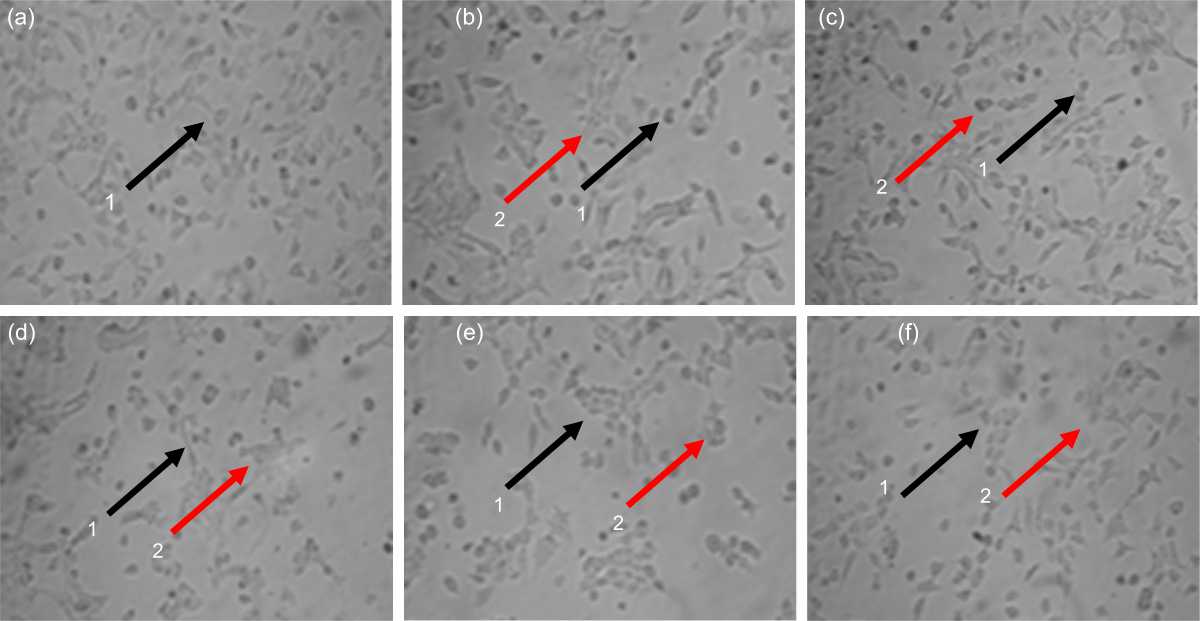
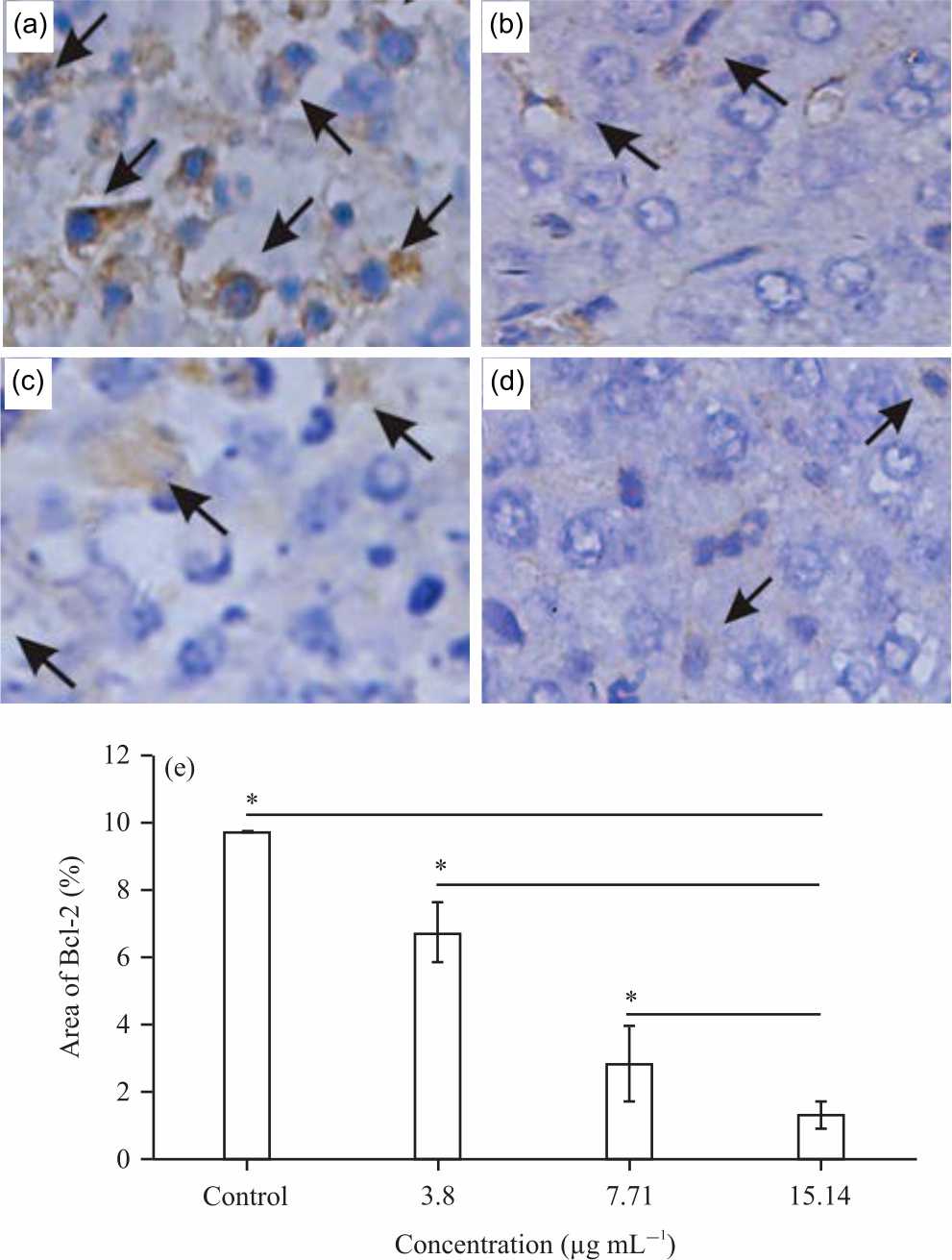
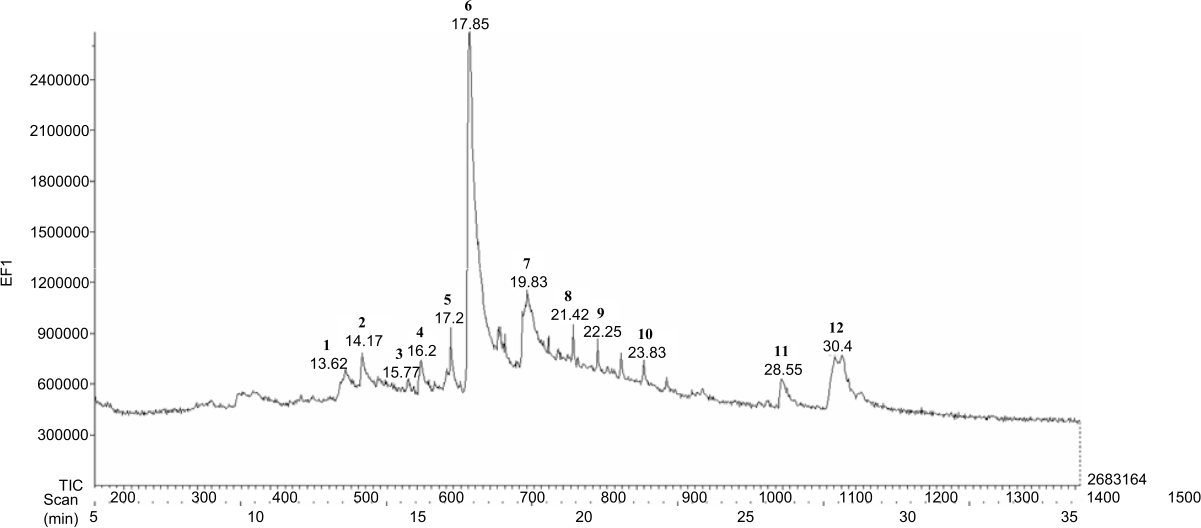
Pterygota alata (Roxb.) R.Br. Bark Fraction Induced Intrinsic Apoptotic Pathway in 4T1 Cells by Decreasing Bcl-2 and Inducing Bax Expression
Department of Chemistry, Faculty of Mathematics and Natural Sciences, Brawijaya University, Malang 65145, Indonesia
LiveDNA: 62.33120
Warsito Warsito
Department of Chemistry, Faculty of Mathematics and Natural Sciences, Brawijaya University, Malang 65145, Indonesia
Masruri Masruri
Department of Chemistry, Faculty of Mathematics and Natural Sciences, Brawijaya University, Malang 65145, Indonesia
Widodo Widodo
Department of Biology, Faculty of Mathematics and Natural Sciences, Brawijaya University, Malang 65145, Indonesia