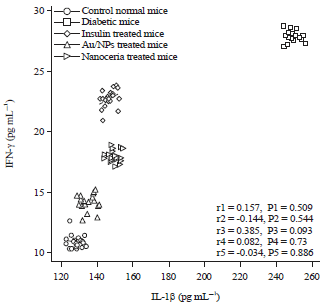
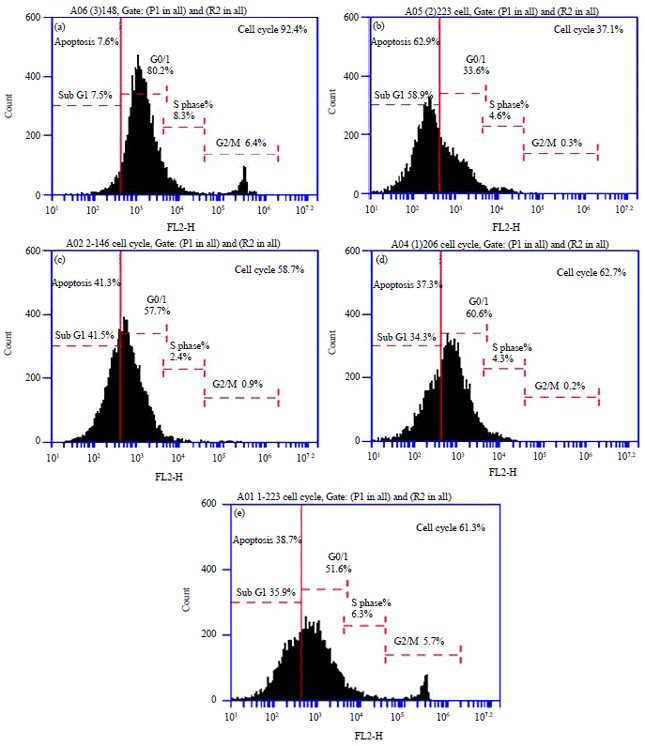
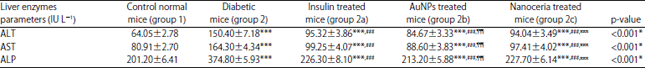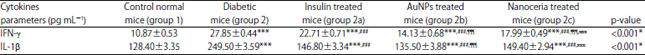Research Article
Effects of Gold and Cerium Oxide Nanoparticles on Type 1 Diabetes in Experimental Mice
Department of Pediatric, Faculty of Medicine, Al Baha University, Al Baha, Saudi Arabia
Mohamed F. El-Refaei
Department of Biochemistry, Faculty of Medicine, Al Baha University, Al Baha, Saudi Arabia
LiveDNA: 20.32261