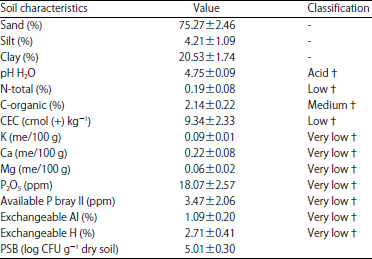
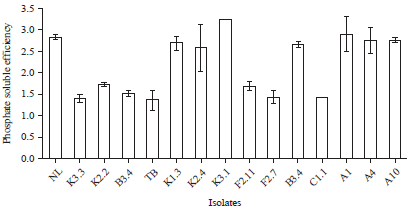
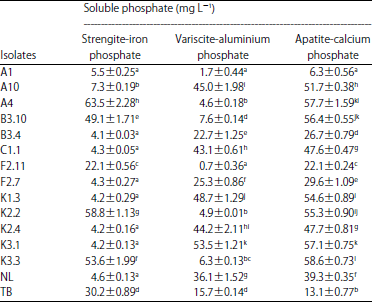
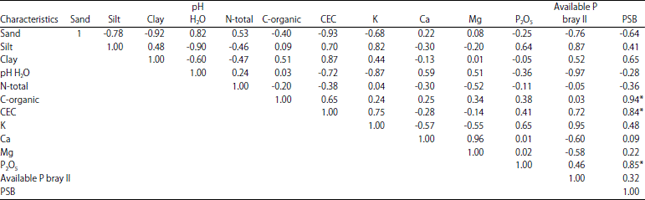
Research Article
Isolation and Solubilisation of Inorganic Phosphate by Burkholderia spp. from the Rhizosphere of Oil Palm
Department of Research and Development, PT Sampoerna Agro Tbk, Jl. Basuki Rahmat No. 788, Palembang, 30127 South Sumatra, Indonesia
LiveDNA: 62.31180
Gregorius Baskara
Department of Research and Development, PT Sampoerna Agro Tbk, Jl. Basuki Rahmat No. 788, Palembang, 30127 South Sumatra, Indonesia
Ruli Wandri
Department of Research and Development, PT Sampoerna Agro Tbk, Jl. Basuki Rahmat No. 788, Palembang, 30127 South Sumatra, Indonesia
Dwi Asmono
Department of Research and Development, PT Sampoerna Agro Tbk, Jl. Basuki Rahmat No. 788, Palembang, 30127 South Sumatra, Indonesia