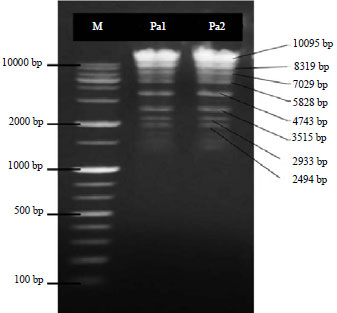
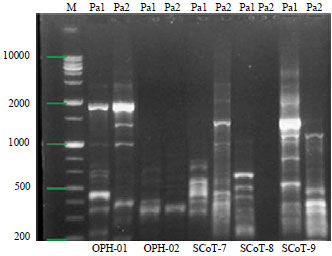
Research Article
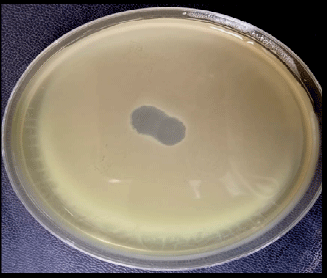
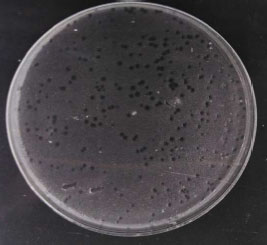
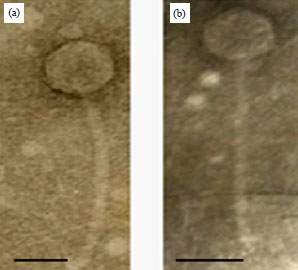
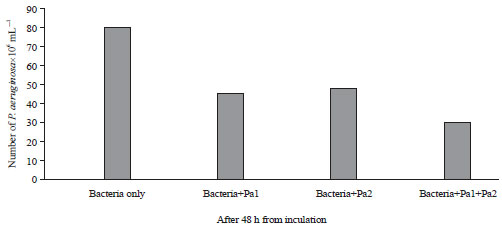
Isolation and Characterization of Pseudomonas aeruginosa and its Virulent Bacteriophages
Department of Agricultural Microbiology, Virology Laboratory, Faculty of Agriculture, Ain Shams University, P.O. Box 68, 11241 Hadayek-Shoubra, Cairo, Egypt
LiveDNA: 20.23985