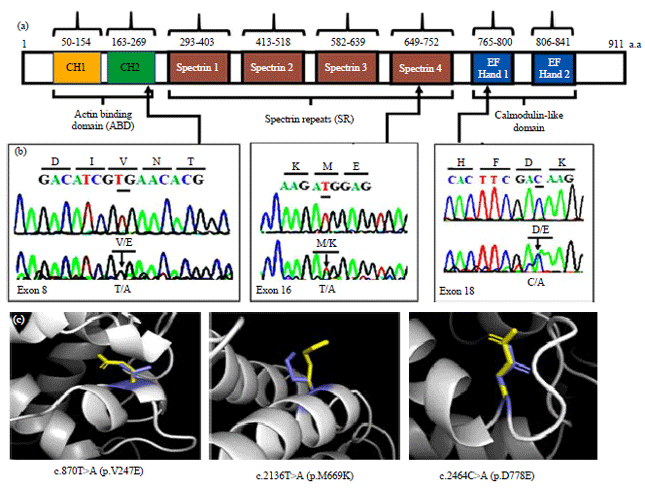
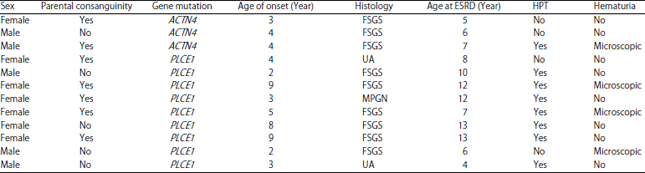
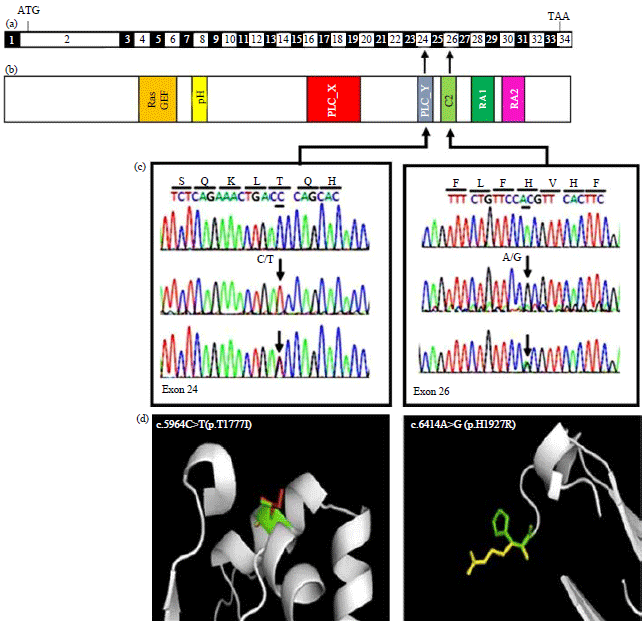
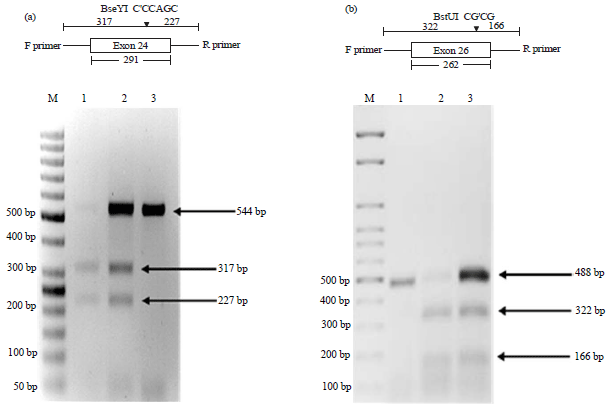
Research Article
Screening of ACTN4 and PLCE1 Genes Mutations in Saudi Children Patients with Steroid Resistant Nephrotic Syndrome
Department of Biology, Faculty of Science, Taif University, P.O. Box 888, Zip code 21974, Taif, Kingdom of Saudi Arabia
LiveDNA: 20.25440
Abdulla A. Alharthi
Department of Pediatrics, Faculty of Medicine, Taif University, Taif, Saudi Arabia
Mohamed M. Hassan
Department of Biology, Faculty of Science, Taif University, P.O. Box 888, Zip code 21974, Taif, Kingdom of Saudi Arabia
Ehab I. El-Hallous
Department of Biology, Faculty of Science, Taif University, P.O. Box 888, Zip code 21974, Taif, Kingdom of Saudi Arabia
Mohamed W. Abukhatwah
Department of Pediatrics, Alhada Armed Forces Hospital, Taif, Saudi Arabia
Abeer M. Almalki
Department of Pediatrics, Children Hospital, Taif, Saudi Arabia
Abdullah A. Muzallef
Abha Maternity Children Hospital, Abha, Saudi Arabia
Saqer Alotaibi
Department of Biotechnology, Faculty of Sciences, Taif University, Taif, Saudi Arabia
LiveDNA: 966.26553
Bandar Aljuaid
Department of Biotechnology, Faculty of Sciences, Taif University, Taif, Saudi Arabia
Walaa F. Alsanie
Department of Clinical Laboratories, Faculty of Applied Medical Sciences, Taif University, Saudi Arabia