Research Article
Role of Human PTEN and TP53 Sequence Mutations in the Etiology of Breast Cancer in Saudi Patients
Faculty of Science, Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia
LiveDNA: 249.31400
Asma S. Algebaly
Faculty of Science, Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia
LiveDNA: 966.16888
Wedad Saeed Alqahtani
Naif Arab University for Security Sciences, Riyadh, Saudi Arabia











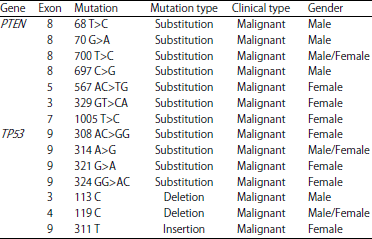
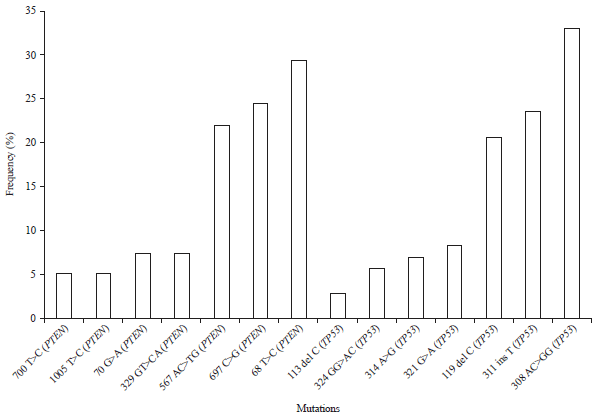
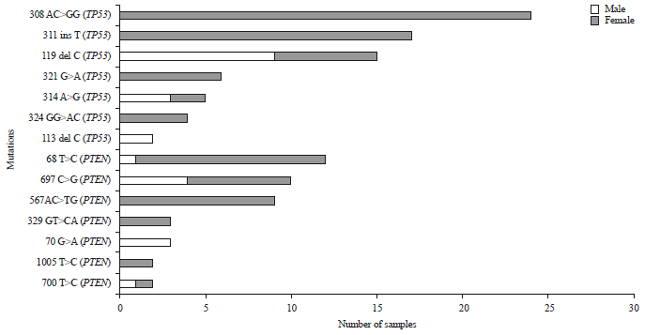
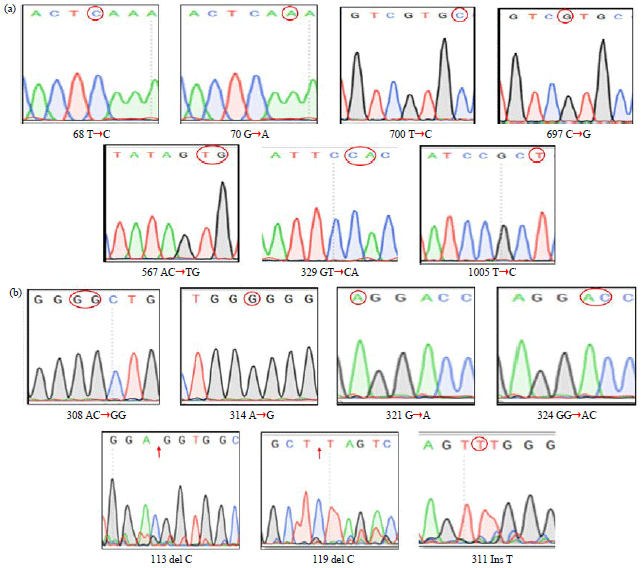

sara ahmed Reply
very interesting abstract.
Mohammad Rasheed Reply
Hello Sir,
I am trying to get pdf of this article but failed its stating bad gateway. I just want to verify the author's affiliations. I will be grateful if you kindly help me. Thank you
Editor
Dear Rasheed,
Thank you for reaching out and bringing this issue to my attention. I apologize for any inconvenience you experienced while trying to access the PDF. I am glad to inform you that the issue has been resolved, and the PDF of the article is now available for download.
You can now access the article and verify the author affiliations. If you have any further questions or need assistance, please feel free to let me know. I am here to help.
Thank you for your patience