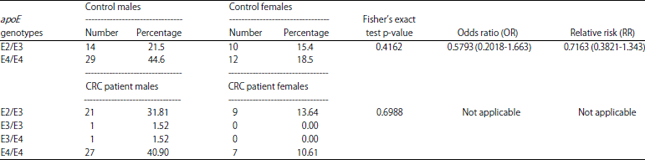
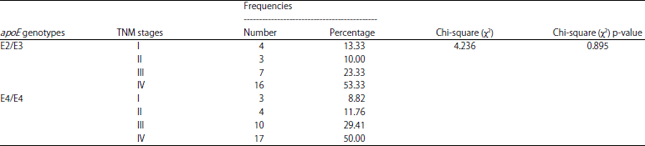
Research Article
Contribution of Apolipoprotein E Isoforms to Colorectal Cancer Carcinogenesis
Department of Biochemistry, Faculty of Science, King Abdulaziz University, Jeddah, Saudi Arabia
Ayat B. Al-Ghafari
Department of Biochemistry, Faculty of Science, King Abdulaziz University, Jeddah, Saudi Arabia
LiveDNA: 966.12767
Huda A. Al Doghaither
Department of Biochemistry, Faculty of Science, King Abdulaziz University, Jeddah, Saudi Arabia
LiveDNA: 966.12928
Huda F. Alshaibi
Department of Biochemistry, Faculty of Science, King Abdulaziz University, Jeddah, Saudi Arabia
Ulfat M. Omar
Department of Biochemistry, Faculty of Science, King Abdulaziz University, Jeddah, Saudi Arabia