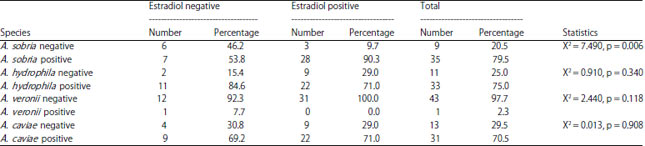
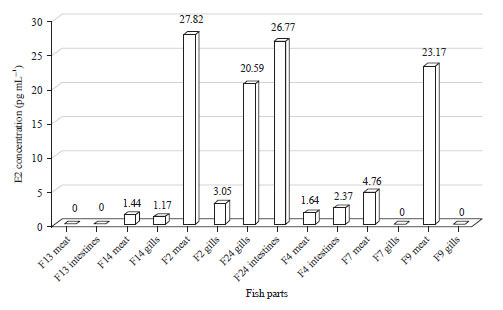
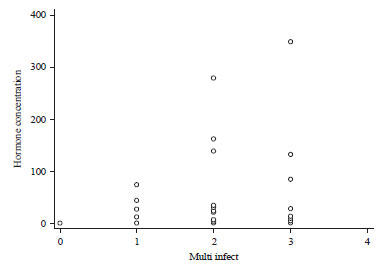
Research Article
Distribution and Potential Effects of 17β-Estradiol (E2) on Aeromonas Diversity in Wastewater and Fish Samples
School of Mathematical and Natural Science, Department of Microbiology, University of Venda, Private Bag X5050, 0950 Thohoyandou, South Africa
Alfred Sichilima
School of Mathematics and Natural Sciences, Department of Biological Sciences, Copperbelt University, P.O. Box 21692, Kitwe, Zambia
Amidou Samie
School of Mathematical and Natural Science, Department of Microbiology, University of Venda, Private Bag X5050, 0950 Thohoyandou, South Africa
LiveDNA: 237.16877