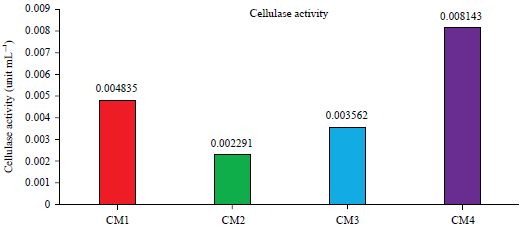
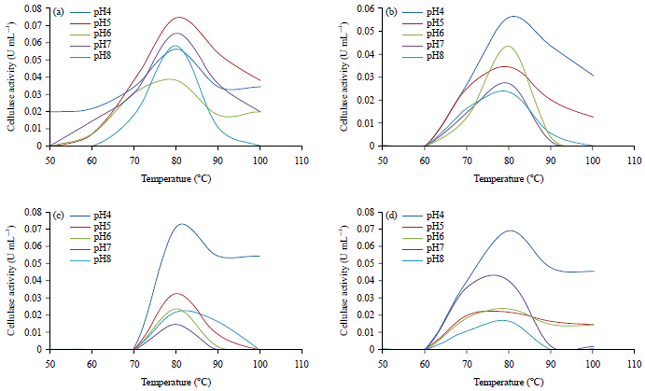
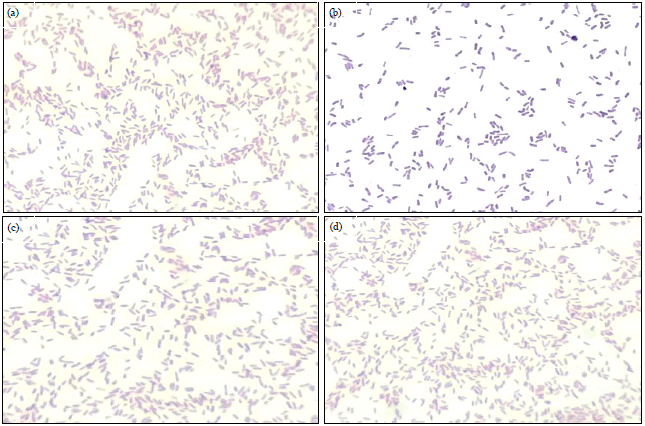
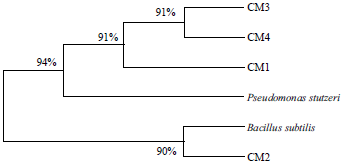
Short Communication
Pseudomonas stutzeri CM1, Novel Thermotolerant Cellulase- Producing Bacteria Isolated from Forest Soil
Major of General Science, Department of Science and Technology, Faculty of Liberal Arts and Science, Roi Et Rajabhat University, Selaphum, Roi Et 4510, Thailand
LiveDNA: 66.12978
Apisit Songsaeng
School of Biotechnology, Institute of Agricultural Technology, Suranaree University of Technology, Muang, Nakhon Ratchasima 30000, Thailand
Thanatip Sriwarom
Major of Biology, Department of Science and Technology, Faculty of Liberal Arts and Science, Roi Et Rajabhat University, Selaphum, Roi Et 4510, Thailand