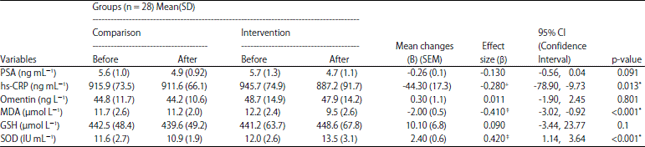
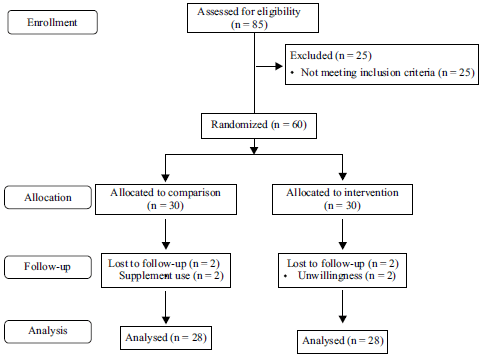
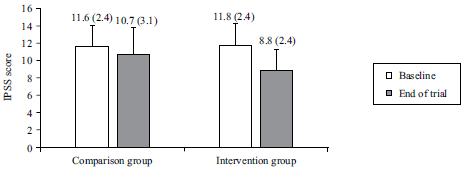
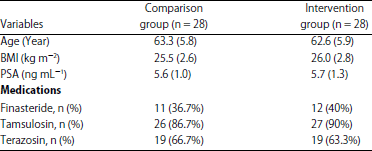Research Article
Urtica Dioica Root Extract on Clinical and Biochemical Parameters in Patients with Benign Prostatic Hyperplasia, Randomized Controlled Trial
Metabolic Diseases Research Center, Research Institute for Prevention of Non-Communicable Diseases, Qazvin University of Medical Sciences, Qazvin, Iran
Mahsa Sheikhsoleimani
Student Research Committee, Qazvin University of Medical Sciences, Qazvin, Iran
Mohammad Reza Memarzadeh
Medicinal Plants Research Center of Barij, Kashan, Iran
Elnaz Haddadi
Student Research Committee, Qazvin University of Medical Sciences, Qazvin, Iran
Mohammad Bakhshpour
Student Research Committee, Qazvin University of Medical Sciences, Qazvin, Iran
Navid Mohammadi
Metabolic Diseases Research Center, Research Institute for Prevention of Non-Communicable Diseases, Qazvin University of Medical Sciences, Qazvin, Iran
Seyyed Mehdi Mirhashemi
Metabolic Diseases Research Center, Research Institute for Prevention of Non-Communicable Diseases, Qazvin University of Medical Sciences, Qazvin, Iran
LiveDNA: 98.28628