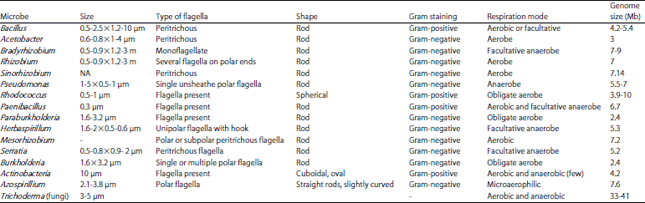
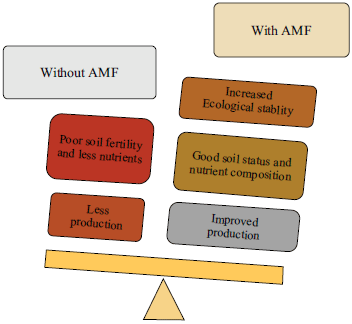
Review Article
Significance of Arbuscular Mycorrhizal Fungi for Acacia: A Review
Department of Botany, Kurukshetra University Kurukshetra, 136119 Kurukshetra, India
Himanshi
Department of Botany, Kurukshetra University Kurukshetra, 136119 Kurukshetra, India
Kavita Rani
Department of Botany, Kurukshetra University Kurukshetra, 136119 Kurukshetra, India
Neha Gill
Department of Botany, Kurukshetra University Kurukshetra, 136119 Kurukshetra, India
Kajal Sandhu
International Center for Genetic Engineering and Biotechnology, Aruna Asaf Ali Marg, New Delhi 110 067, India
Neema Bisht
International Center for Genetic Engineering and Biotechnology, Aruna Asaf Ali Marg, New Delhi 110 067, India
Tarun Kumar
Department of Forestry, Chaudhary Charan Singh Haryana Agricultural University, 125004 Hisar, India
LiveDNA: 91.16284
Prashant Kaushik
Instituto de Conservaci�n y Mejora de la Agrodiversidad Valenciana, Universitat Polit�cnica de Val�ncia, 46022 Valencia, Spain
LiveDNA: 91.32045