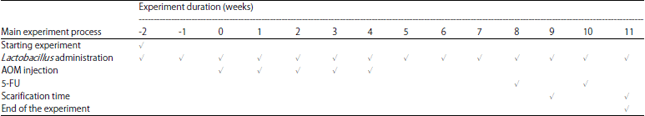
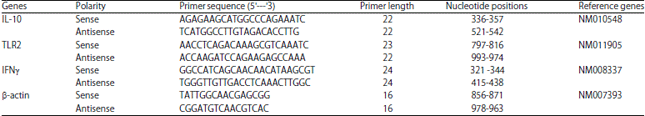
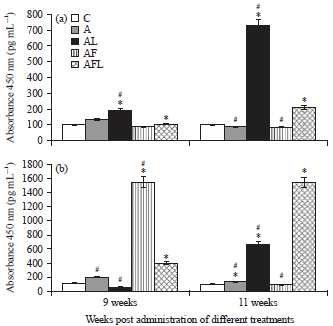
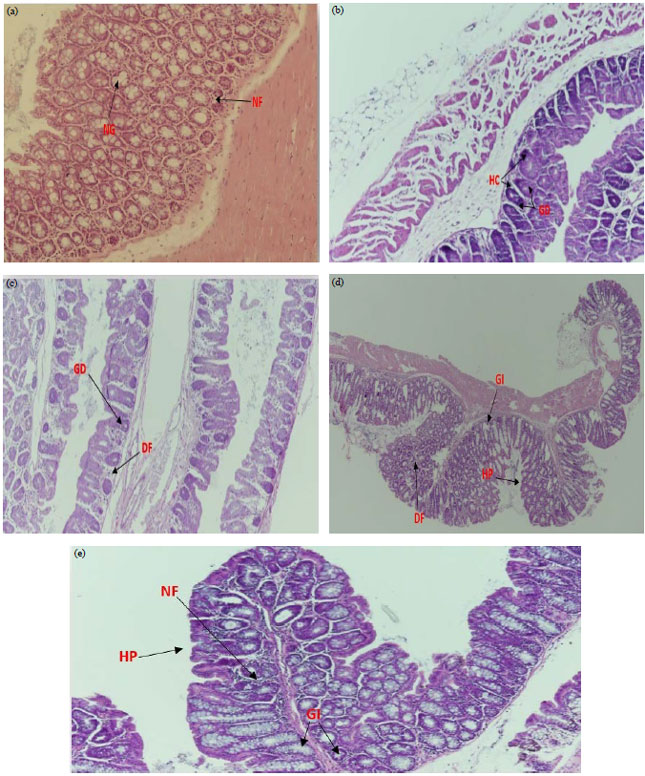
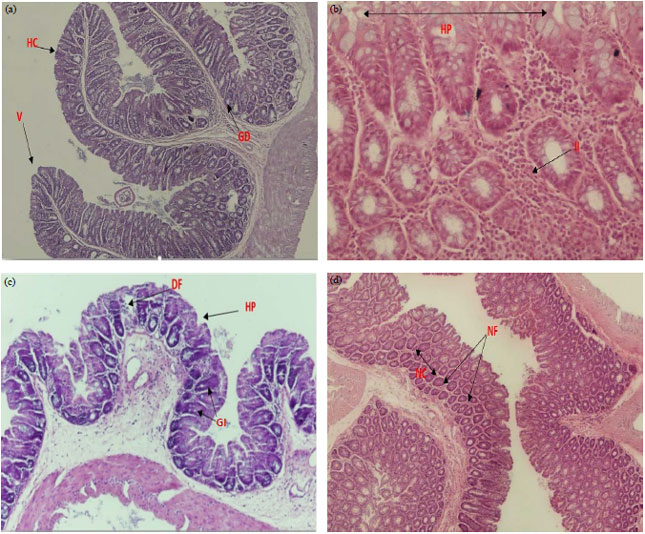
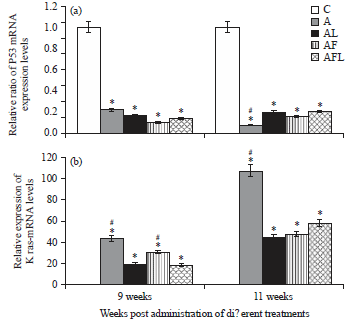
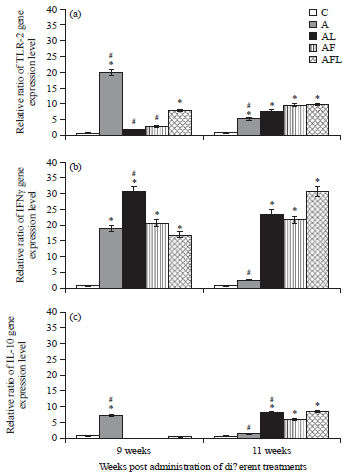
Research Article
Lactobacillus rhamnosus Enhances the Immunological Antitumor Effect of 5-Fluorouracil against Colon Cancer
Department of Biological Science, Faculty of Science, King Abdulaziz University, Jeddah, Kingdom of Saudi Arabia
LiveDNA: 20.7590
Boshra Al Hazmi
Department of Biological Science, Faculty of Science, King Abdulaziz University, Jeddah, Kingdom of Saudi Arabia
Alawiah Alhebshi
Department of Biological Science, Faculty of Science, King Abdulaziz University, Jeddah, Kingdom of Saudi Arabia
Alia M. Aldahlawi
Department of Biological Science, Faculty of Science, King Abdulaziz University, Jeddah, Kingdom of Saudi Arabia
Reem Al Bassam
Department of Histopathology, King Abdulaziz Hospital, Jeddah, Kingdom of Saudi Arabia