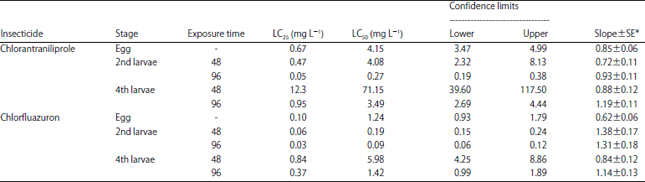
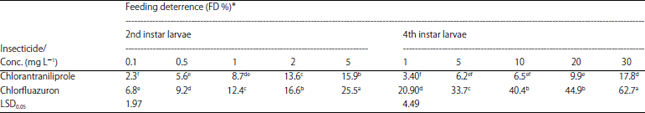
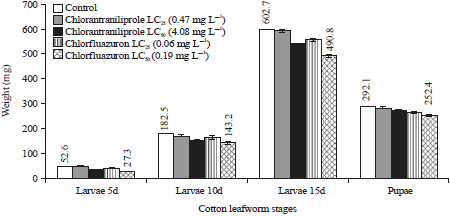
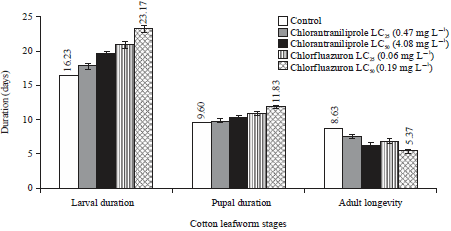
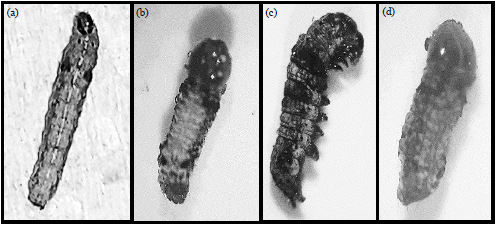
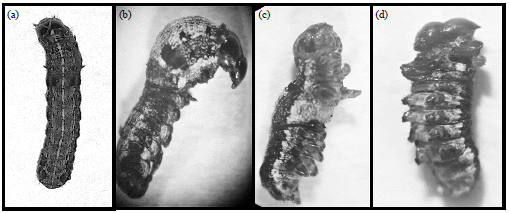
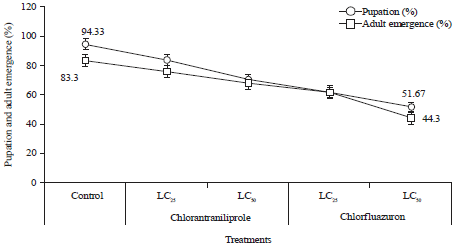
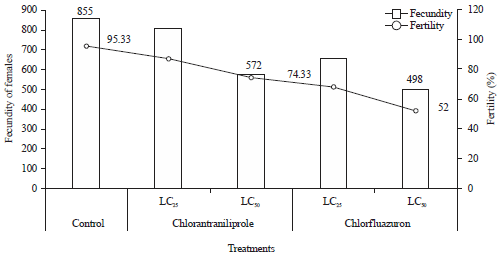
Research Article
Insecticidal, Behavioral and Biological Effects of Chlorantraniliprole and Chlorfluazuron on Cotton Leafworm (Spodoptera littoralis)
Department of Applied Entomology and Zoology, Faculty of Agriculture, Alexandria University, Alexandria, Egypt
LiveDNA: 20.16282
Sahar Elsayed Eldesouky
Department of Cotton Pesticides Evaluation, Plant Protection Research Institute, Agricultural Research Center, El-Sabhia, Alexandria, Egypt