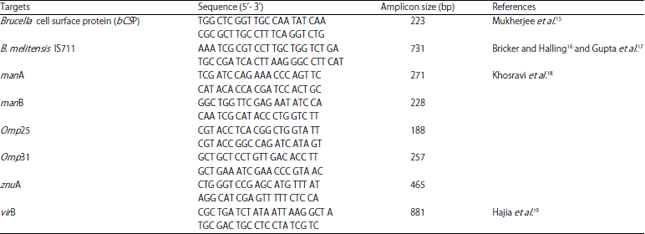
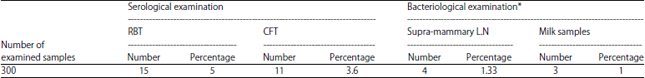
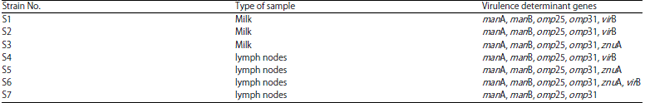
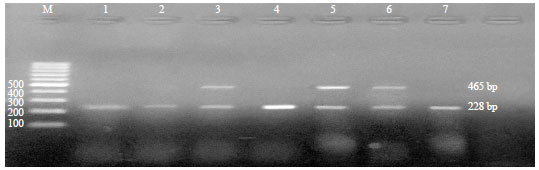
Research Article
Substantial Virulence Genes among Brucella melitensis Field Strains Isolated from Cattle in Egypt
Laboratory of Brucella, Department of Reproductive Diseases, Animal Reproduction Research Institute, Giza, Egypt
LiveDNA: 20.15017
Walid S. Mousa
Department of Animal Medicine and Infectious Diseases, Faculty of Veterinary Medicine, University of Sadat City, Egypt
LiveDNA: 20.27367
Jehan A. Gafer
Biotechnology Unit, Animal Reproduction Research Institute, Giza, Egypt
Hamed T. Elbaz
Department of Theriogenology, Faculty of Veterinary Medicine, University of Sadat City, Egypt
Eman Abdeen
Department of Bacteriology, Mycology and Immunology, Faculty of Veterinary Medicine, University of Sadat City, Egypt
Heba Hussien
Department of Food Hygiene and Control, Faculty of Veterinary Medicine, University of Sadat City, Egypt