Research Article
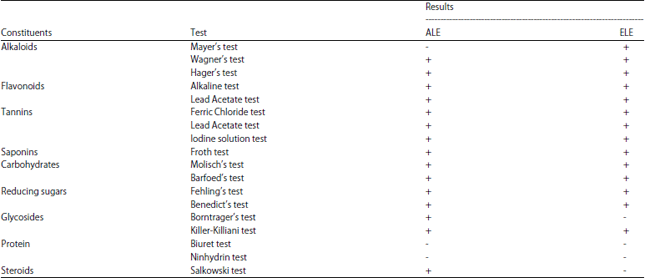
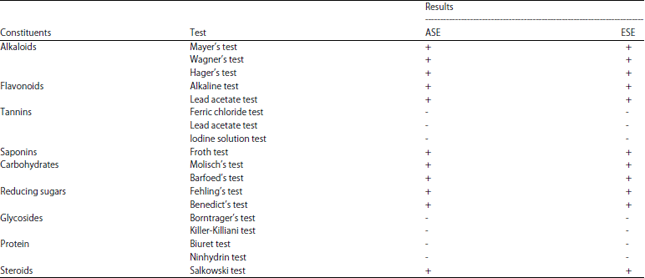
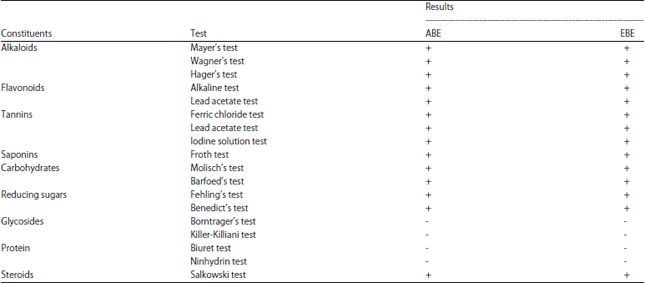
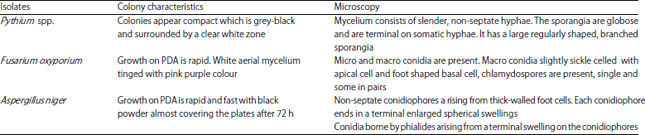
Inhibitory Effect of Aqueous and Ethanolic Extracts of Neem Parts on Fungal Rot Disease of Solanum tuberosum
Department of Biochemistry, Faculty of Pure and Applied Sciences, Federal University Wukari, Nigeria
Verwiyeh Silas Tatah
Department of Biochemistry, Faculty of Pure and Applied Sciences, Federal University Wukari, Nigeria
Chinedu Imo
Department of Biochemistry, Faculty of Pure and Applied Sciences, Federal University Wukari, Nigeria
LiveDNA: 234.23829
Emmanuel Mamma
Department of Biochemistry, Faculty of Pure and Applied Sciences, Federal University Wukari, Nigeria
Mida Habila Mayel
Department of Biochemistry, Faculty of Pure and Applied Sciences, Federal University Wukari, Nigeria
Ayobami Jahdahunsi Kukoyi
Department of Biochemistry, Faculty of Pure and Applied Sciences, Federal University Wukari, Nigeria
Ibrahim Adams Jeji
Department of Biochemistry, Faculty of Pure and Applied Sciences, Federal University Wukari, Nigeria