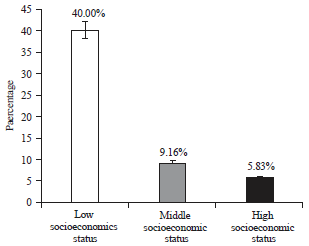
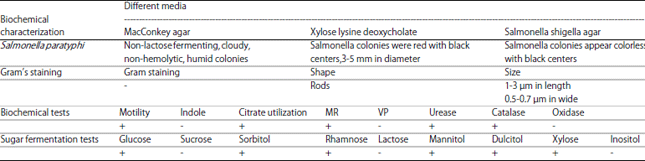
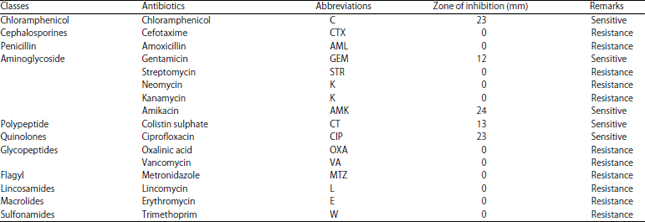
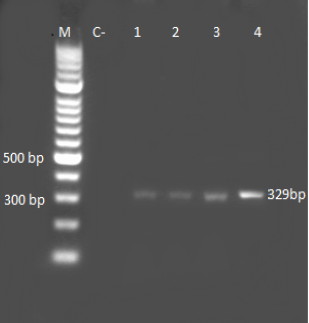
Research Article
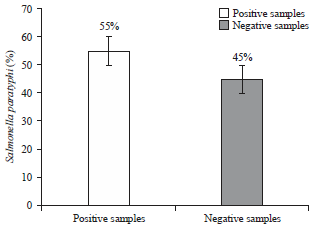
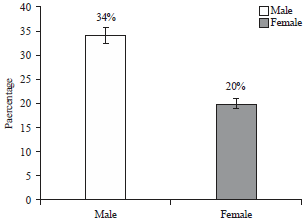
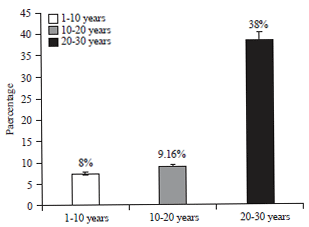
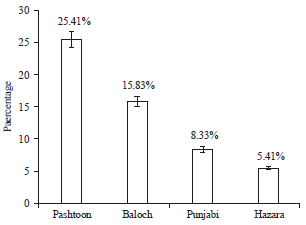
Isolation and Identification of Salmonella paratyphi from Enteric Fever Patients at Different Hospitals of Quetta City
Department of Microbiology, University of Balochistan, Quetta, Balochistan, Pakistan
Muhammad Kamran Taj
Center for Advance Studies in Vaccinology and Biotechnology, University of Balochistan, Quetta, Pakistan
LiveDNA: 92.24011
Imran Nawaz
Department of Microbiology, University of Balochistan, Quetta, Balochistan, Pakistan
Imran Taj
Center for Advance Studies in Vaccinology and Biotechnology, University of Balochistan, Quetta, Pakistan
Marina Panezai
institute of Biochemistry, University of Balochistan, Quetta, Pakistan
Nazia Panezai
Department of Microbiology, University of Balochistan, Quetta, Balochistan, Pakistan
Umbreen Zafar
Department of Microbiology, University of Balochistan, Quetta, Balochistan, Pakistan
LiveDNA: 92.23518
Durdana Ghulam Muhammad
Department of Microbiology, University of Balochistan, Quetta, Balochistan, Pakistan
Saeed Ahmed Essote
Department of Zoology, University of Balochistan, Quetta, Pakistan
Ghulam Muhammad
Center for Advance Studies in Vaccinology and Biotechnology, University of Balochistan, Quetta, Pakistan