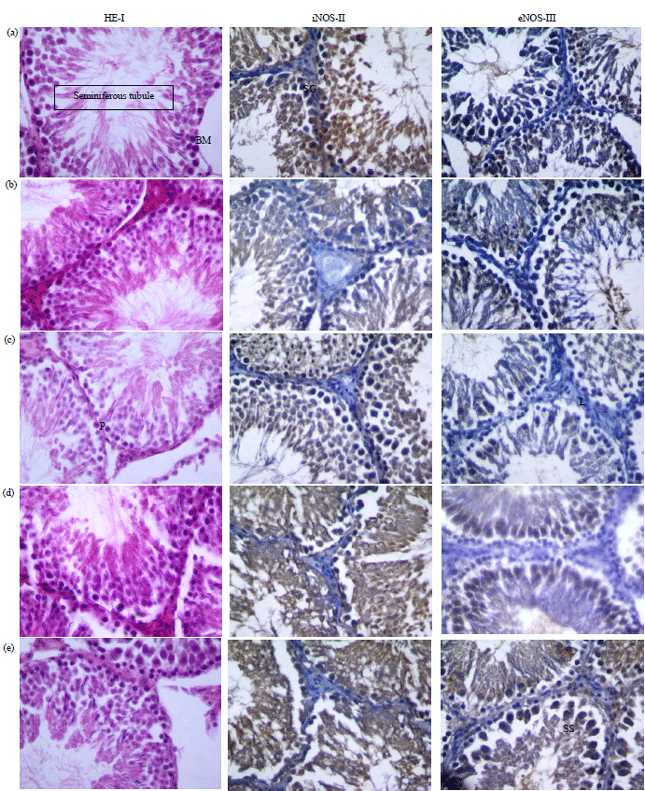
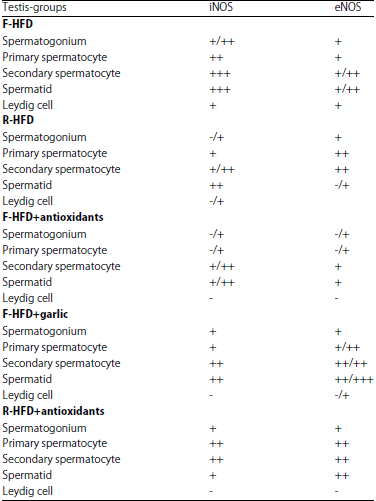
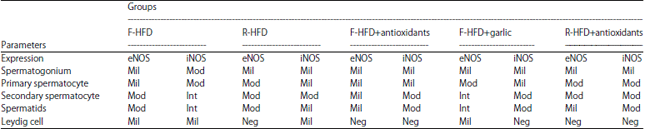
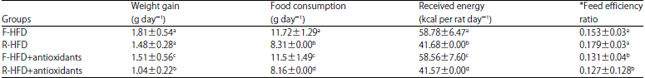Research Article
Effect of Antioxidants on Testicular iNOS and eNOS after High-Fat Diet in Rat
Department of Anatomical Sciences, Faculty of Medicine, Hamadan University of Medical Sciences, Shahid Fahmideh Street, 65178-3-8736 Hamadan, Iran
LiveDNA: 98.16590
Mahnaz Hosseini
Department of Anatomical Sciences, Faculty of Medicine, Hamadan University of Medical Sciences, Shahid Fahmideh Street, 65178-3-8736 Hamadan, Iran
Sevinc Inan
Department of Histology and Embryology, Faculty of Medicine, Celal Bayar University, Manisa, Turkey
Zohreh Alizadeh
Department of Anatomical Sciences, Faculty of Medicine, Hamadan University of Medical Sciences, Shahid Fahmideh Street, 65178-3-8736 Hamadan, Iran
LiveDNA: 98.14007
Mehrangiz Vahabian
Department of Anatomical Sciences, Faculty of Medicine, Hamadan University of Medical Sciences, Shahid Fahmideh Street, 65178-3-8736 Hamadan, Iran
LiveDNA: 98.13739
Ali Asghar Vahidinia
Department of Biochemistry and Nutrition, Faculty of Medicine, Hamadan University of Medical Sciences, Shahid Fahmideh Street, 65178-3-8736 Hamadan, Iran
Hosein Lahoutian
Department of Anatomical Sciences, Faculty of Medicine, Hamadan University of Medical Sciences, Shahid Fahmideh Street, 65178-3-8736 Hamadan, Iran