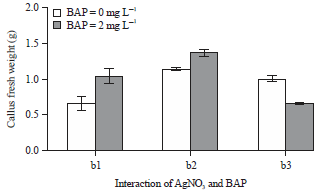
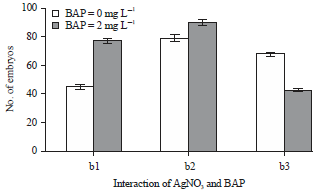
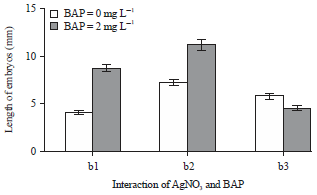
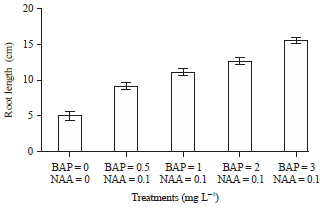
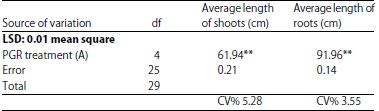
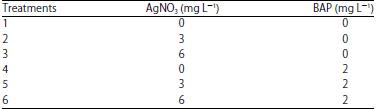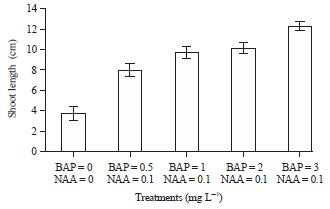Research Article
Effect of AgNO3 and BAP on Root as a Novel Explant in Date Palm (Phoenix dactylifera cv. Medjool) Somatic Embryogenesis
Faculty of Agricultural Sciences, University of Guilan, Rasht, Iran
Reza Zarghami
Agricultural Biotechnology Research Institute of Iran (ABRII), Karaj, Iran
Hassan Hassani
Faculty of Agricultural Sciences, University of Guilan, Rasht, Iran
Hedayat Zakizadeh
Faculty of Agricultural Sciences, University of Guilan, Rasht, Iran
Ali Salari
National Research Center on Plant Biotechnology (NRCPB), New Delhi, India