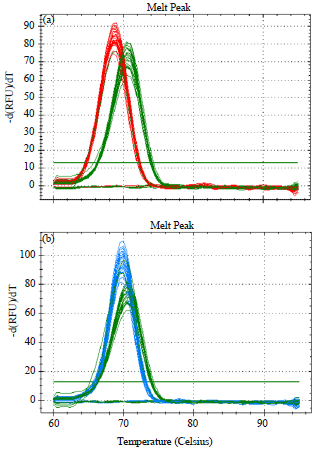
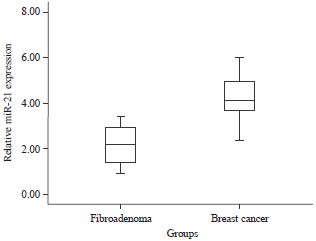
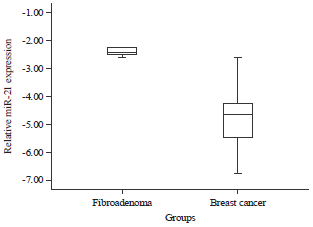
Research Article
Evaluation of MiR-21 and MiR-10b Expression of Human Breast Cancer in West Sumatera
Department of Anatomy, Faculty of Medicine, Andalas University, Padang, Indonesia
Wirsma A. Harahap
Division of Surgical Oncology, Medical School of M. Djamil Hospital, Andalas University, Padang, Indonesia
Dessy Arisanty
Department of Biochemistry, Faculty of Medicine, Andalas University, Padang, Indonesia