Research Article
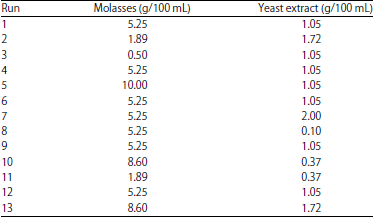
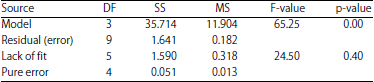
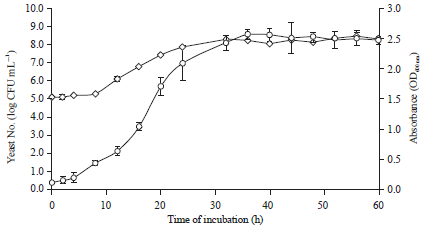
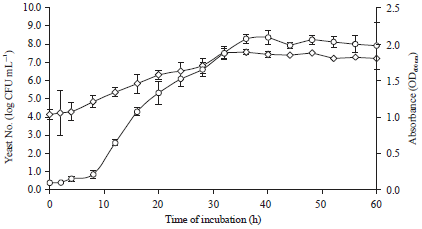
Optimization of Culture Medium for the Growth of Candida sp. and Blastobotrys sp. as Starter Culture in Fermentation of Cocoa Beans (Theobroma cacao) Using Response Surface Methodology (RSM)
Department of Food Science, Faculty of Food Science and Technology,
Z. Zakuan
Department of Food Science, Faculty of Food Science and Technology,
H. Norhayati
Department of Food Technology, Faculty of Food Science and Technology,
A.S. MeorHussin
Department of Food Technology, Faculty of Food Science and Technology,
Y. Rukayadi
Department of Food Science, Faculty of Food Science and Technology,