Research Article
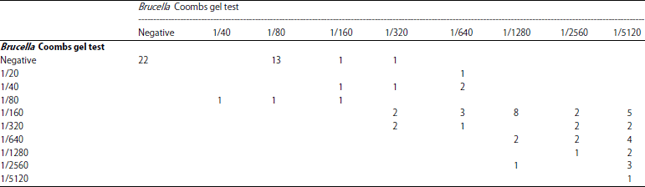
Evaluation of a New and Rapid Serologic Test for Detecting Brucellosis: Brucella Coombs Gel Test
Department of Pharmaceutical Microbiology, Faculty of Pharmacy, Atat�rk University, Erzurum, Turkey
Hakan Igan
Department of Microbiology, Palandoken State Hospital, Erzurum, Turkey
Muhammet Hamidullah Uyanik
Department of Medical Microbiology, Faculty of Medical, Atat�rk University, Erzurum, Turkey