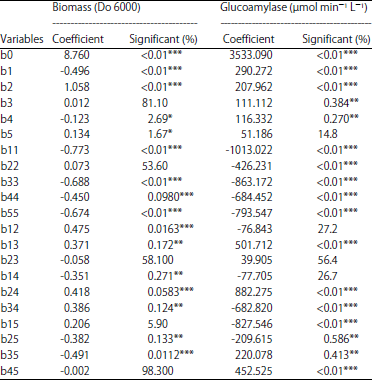
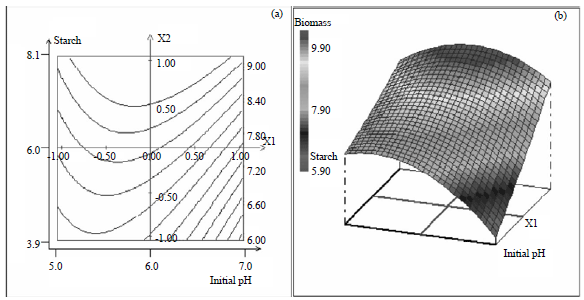
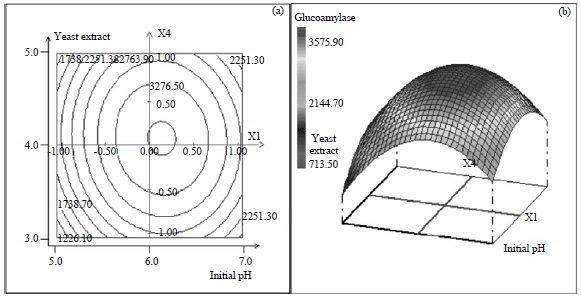
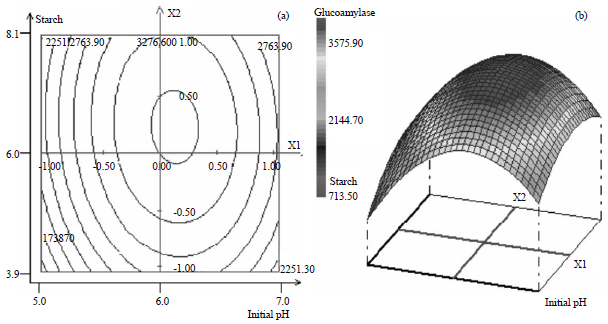
Research Article
Application of Response Surface Methodology for Optimization of Extracellular Glucoamylase Production by Candida guilliermondii
Laboratory of Agro-physiology, Biotechnologies and Quality, Sciences College, University IBN Tofail, BP 133, 14000 Kenitra, Morocco
Youssfi El Kettani
C.I.R.O.S Laboratory, Department of Mathematics, Sciences College, University IBN Tofail, BP 133, 14000 Kenitra, Morocco
Aitounejjar Ali
National Institute of Agronomic Research, P.O. Box 589, Settat 26000, Morocco
Elyachioui Mohamed
Laboratory of Agro-physiology, Biotechnologies and Quality, Sciences College, University IBN Tofail, BP 133, 14000 Kenitra, Morocco
Jadal Mohamed
Laboratory of Agro-physiology, Biotechnologies and Quality, Sciences College, University IBN Tofail, BP 133, 14000 Kenitra, Morocco