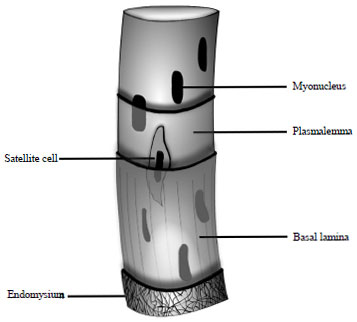
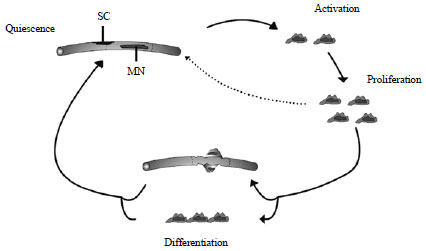
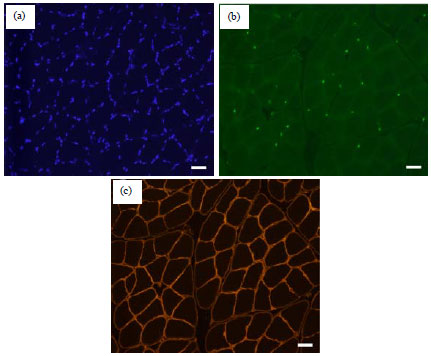
Mini Review
Myogenic Satellite Cells: Biological Milieu and Possible Clinical Applications
Department of Anatomy, Faculty of Medicine, Jordan University of Science and Technology, 22110 Irbid, Jordan
Ayman G. Mustafa
Department of Anatomy, Faculty of Medicine, Jordan University of Science and Technology, 22110 Irbid, Jordan
Hasan A. Asfour
Department of Anatomy, Faculty of Medicine, Jordan University of Science and Technology, 22110 Irbid, Jordan
Emad I. Shaqoura
Department of Anatomy, Faculty of Medicine, Jordan University of Science and Technology, 22110 Irbid, Jordan