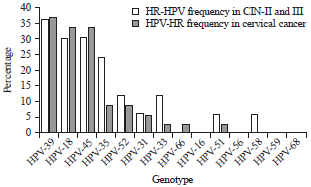
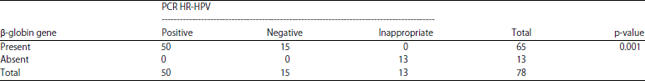
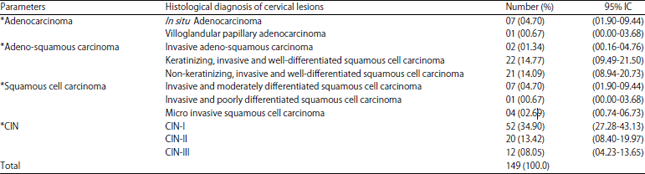
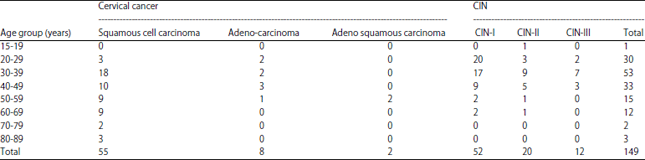
Research Article
Molecular Epidemiology of High-Risk Human Papillomavirus in High-Grade Cervical Intraepithelial Neoplasia and in Cervical Cancer in Parakou, Republic of Benin
Laboratory of Molecular Biology and Genetics (LABIOGENE), University of Ouagadougou, 03 BP 7021 Ouagadougou 03, Burkina Faso Pietro Annigoni Biomolecular Research Center (CERBA), 01 BP 364 Ouagadougou 01, Burkina Faso Teaching and Research Unit in Biochemistry and Molecular Biology, Faculty of Medicine, University of Parakou, BP 123 Parakou, Republic of Benin Faculty of Medicine, University of Saint Thomas dAquin, 06 BP 10212 Ouagadougou 01, Burkina Faso
T.C. Ouedraogo
Pietro Annigoni Biomolecular Research Center (CERBA), 01 BP 364 Ouagadougou 01, Burkina Faso Teaching and Research Unit in Biochemistry and Molecular Biology, Faculty of Medicine, University of Parakou, BP 123 Parakou, Republic of Benin
L.V.C. Brun
Teaching and Research Unit in Biochemistry and Molecular Biology, Faculty of Medicine, University of Parakou, BP 123 Parakou, Republic of Benin Teaching and Research Unit, Department of Anatomy and Cytopathology, Faculty of Medicine, University of Parakou, BP 123 Parakou, Republic of Benin
D. Obiri-Yeboah
Department of Microbiology, School of Medical Sciences, University of Coast, University Post Office, Ghana
W.F. Djigma
Laboratory of Molecular Biology and Genetics (LABIOGENE), University of Ouagadougou, 03 BP 7021 Ouagadougou 03, Burkina Faso Pietro Annigoni Biomolecular Research Center (CERBA), 01 BP 364 Ouagadougou 01, Burkina Faso
S. Kabibou
Department of Service of Gynecology, University Hospital of Borgou and Alibori (CHUD-B/A), BP 123 Parakou, Republic of Benin
S. Ouattara
Yalgado Ouedraogo University Hospital (CHU/YO), 03 BP 7022, Republic of Benin
M. Gomina
Teaching and Research Unit in Biochemistry and Molecular Biology, Faculty of Medicine, University of Parakou, BP 123 Parakou, Republic of Benin
A.T. Yonli
Laboratory of Molecular Biology and Genetics (LABIOGENE), University of Ouagadougou, 03 BP 7021 Ouagadougou 03, Burkina Faso Pietro Annigoni Biomolecular Research Center (CERBA), 01 BP 364 Ouagadougou 01, Burkina Faso
V.J.T.E. Bazie
Laboratory of Molecular Biology and Genetics (LABIOGENE), University of Ouagadougou, 03 BP 7021 Ouagadougou 03, Burkina Faso Pietro Annigoni Biomolecular Research Center (CERBA), 01 BP 364 Ouagadougou 01, Burkina Faso
C. Ouedraogo
Pietro Annigoni Biomolecular Research Center (CERBA), 01 BP 364 Ouagadougou 01, Burkina Faso Yalgado Ouedraogo University Hospital (CHU/YO), 03 BP 7022, Republic of Benin
O. Lompo
Yalgado Ouedraogo University Hospital (CHU/YO), 03 BP 7022, Republic of Benin
S.A. Akpona
Pietro Annigoni Biomolecular Research Center (CERBA), 01 BP 364 Ouagadougou 01, Burkina Faso Teaching and Research Unit in Biochemistry and Molecular Biology, Faculty of Medicine, University of Parakou, BP 123 Parakou, Republic of Benin
J. Simpore
Laboratory of Molecular Biology and Genetics (LABIOGENE), University of Ouagadougou, 03 BP 7021 Ouagadougou 03, Burkina Faso Pietro Annigoni Biomolecular Research Center (CERBA), 01 BP 364 Ouagadougou 01, Burkina Faso Faculty of Medicine, University of Saint Thomas dAquin, 06 BP 10212 Ouagadougou 01, Burkina Faso