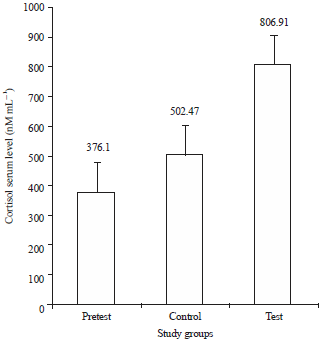
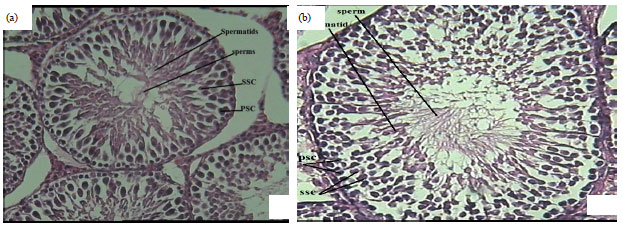
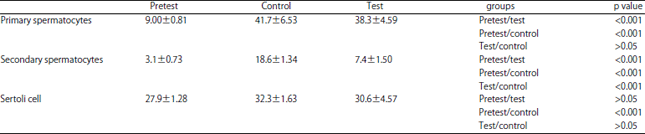
Research Article
Effects of Prepubertal Acute Immobilization Stress on Serum Kisspeptin Level and Testis Histology in Rats
Student Research Committee, Jahrom University of Medical Sciences, Jahrom, Iran
Abdolreza Sotoodeh Jahromi
Research Center for non-Communicable Diseases, Jahrom University of Medical Sciences, Jahrom, Iran
Alireza Yusefi
Research Center for non-Communicable Diseases, Jahrom University of Medical Sciences, Jahrom, Iran
Ali Razeghi
Department of Pathology, Jahrom University of Medical Science, Jahrom, Iran
Hassan Zabetiyan
Research Center for non-Communicable Diseases, Jahrom University of Medical Sciences, Jahrom, Iran
Mohammad Yasin Karami
Department of Surgery, Shiraz University of Medical Science, Shiraz, Iran
Abdol Hossein Madani
Research Center for Social Determinants of Health Promotion, Hormozgan University of Medical Sciences, Bandarabbas, Iran