Research Article
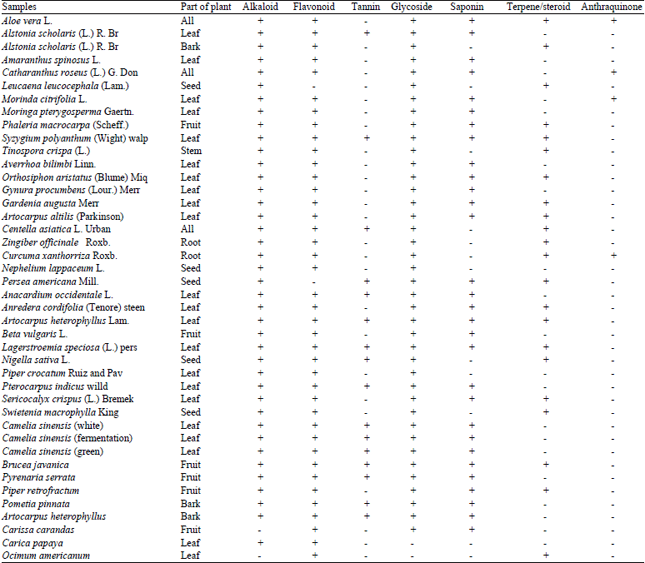
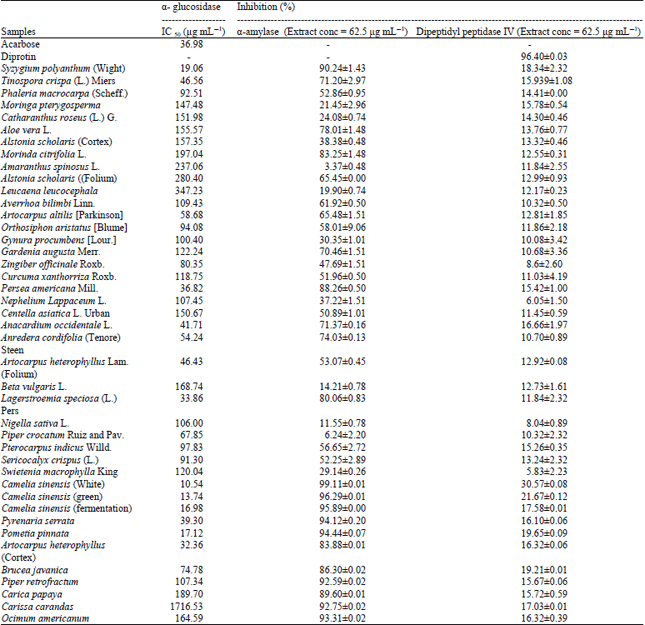
Antidiabetic Activity and Phytochemical Screening of Extracts from Indonesian Plants by Inhibition of Alpha Amylase, Alpha Glucosidase and Dipeptidyl Peptidase IV
Faculty of Pharmacy, University of Indonesia, Postal Code, 16424, Depok, Indonesia
Rosita Handayani
Faculty of Pharmacy, University of Indonesia, Postal Code, 16424, Depok, Indonesia
Rani Sauriasari
Faculty of Pharmacy, University of Indonesia, Postal Code, 16424, Depok, Indonesia
Azizahwati
Faculty of Pharmacy, University of Indonesia, Postal Code, 16424, Depok, Indonesia
Uqie Shabrina Hasyyati
Faculty of Pharmacy, University of Indonesia, Postal Code, 16424, Depok, Indonesia
Idam Titis Permana
Faculty of Pharmacy, University of Indonesia, Postal Code, 16424, Depok, Indonesia
Yunita Indah Permatasari
Faculty of Pharmacy, University of Indonesia, Postal Code, 16424, Depok, Indonesia