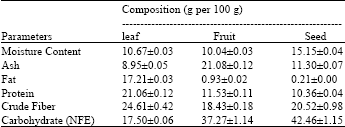
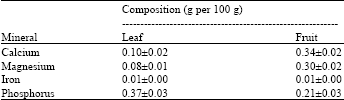
Research Article
Analyses of the Leaf, Fruit and Seed of Thaumatococcus daniellii (Benth.): Exploring Potential Uses
Department of Biological Sciences, School of Applied and Natural Sciences, College of Science and Technology, Covenant University, Canaanland, PMB 1023 Ota, Ogun State, Nigeria
Adetayo Y. Oluwadamisi
Department of Biological Sciences, School of Applied and Natural Sciences, College of Science and Technology, Covenant University, Canaanland, PMB 1023 Ota, Ogun State, Nigeria
Samuel T. Popoola
Department of Biological Sciences, School of Applied and Natural Sciences, College of Science and Technology, Covenant University, Canaanland, PMB 1023 Ota, Ogun State, Nigeria
Bolaji J. David
Department of Biological Sciences, School of Applied and Natural Sciences, College of Science and Technology, Covenant University, Canaanland, PMB 1023 Ota, Ogun State, Nigeria
Tamunotonyesia Epelle
Department of Biological Sciences, School of Applied and Natural Sciences, College of Science and Technology, Covenant University, Canaanland, PMB 1023 Ota, Ogun State, Nigeria