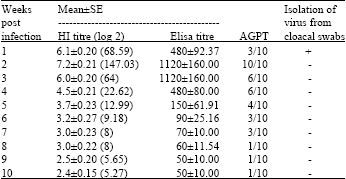
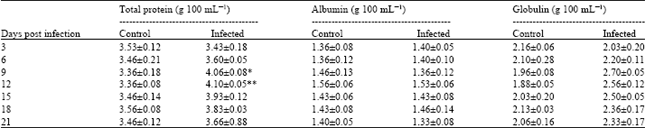
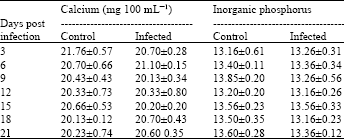
Research Article
Egg Drop Syndrome-76 (EDS-76) in Japanese Quails (Coturnix coturnix japonica): An Experimental Study Revealing Pathology, Effect on Egg Production/Quality and Immune Responses
Avian Diseases Section, Division of Pathology, Indian Veterinary Research Institute, Izatnagar, 243122, Bareilly (UP), India
Jag Mohan Kataria
Avian Diseases Section, Division of Pathology, Indian Veterinary Research Institute, Izatnagar, 243122, Bareilly (UP), India
Sandip Chakraborty
Department of Animal Resources Development, Pt. Nehru Complex, Agartala, Pin, 799006, India
Kuldeep Dhama
Avian Diseases Section, Division of Pathology, Indian Veterinary Research Institute, Izatnagar, 243122, Bareilly (UP), India