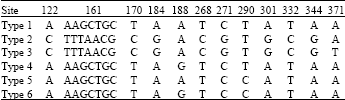
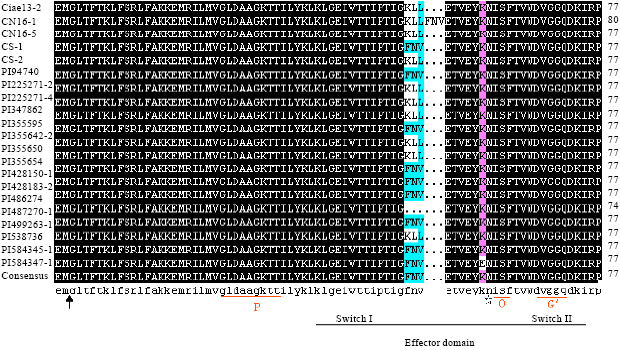
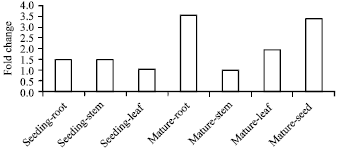
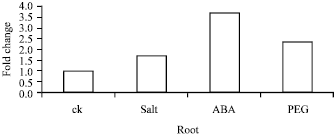
Research Article
Characterization and Chromosome Location of ADP-ribosylation Factors (ARFs) in Wheat
College of Agronomy, Sichuan Agricultural University, Wenjiang, 611130, People�s Republic of China
Guoyue Chen
Triticeae Research Institute, Sichuan Agricultural University, Wenjiang, 611130, People�s Republic of China
Jirui Wang
Triticeae Research Institute, Sichuan Agricultural University, Wenjiang, 611130, People�s Republic of China
Yaxi Liu
Triticeae Research Institute, Sichuan Agricultural University, Wenjiang, 611130, People�s Republic of China
Qiantao Jiang
Triticeae Research Institute, Sichuan Agricultural University, Wenjiang, 611130, People�s Republic of China
Wei Li
College of Agronomy, Sichuan Agricultural University, Wenjiang, 611130, People�s Republic of China
Xiujing Lan
Triticeae Research Institute, Sichuan Agricultural University, Wenjiang, 611130, People�s Republic of China
Shoufen Dai
Triticeae Research Institute, Sichuan Agricultural University, Wenjiang, 611130, People�s Republic of China
Yuming Wei
Triticeae Research Institute, Sichuan Agricultural University, Wenjiang, 611130, People�s Republic of China
Youliang Zheng
Triticeae Research Institute, Sichuan Agricultural University, Wenjiang, 611130, People�s Republic of China