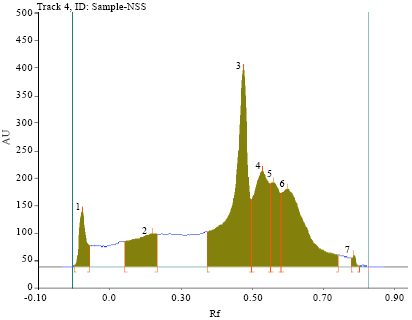
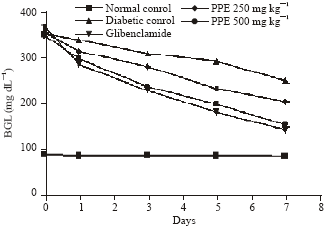
Research Article
Therapeutic Hypoglycemic Potential of Pentapetes phoenicea L. in Experimentally Induced Hyperglycemic Rats
University Institute of Pharmacy, Chhatrapati Shahu Ji Maharaj University, Kalyanpur, Kanpur, 208024, India
Prakash Chandra Gupta
University Institute of Pharmacy, Chhatrapati Shahu Ji Maharaj University, Kalyanpur, Kanpur, 208024, India
Chandana Venkateshwar Rao
Pharmacognosy and Ethnopharmacology Division, National Botanical Research Institute, Lucknow, Uttar Pradesh, India