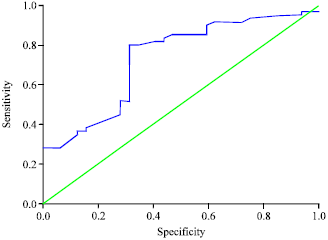
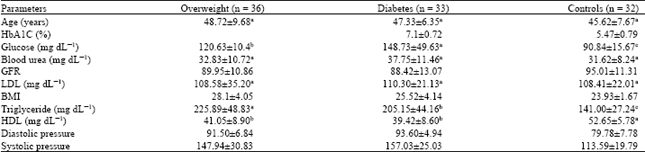
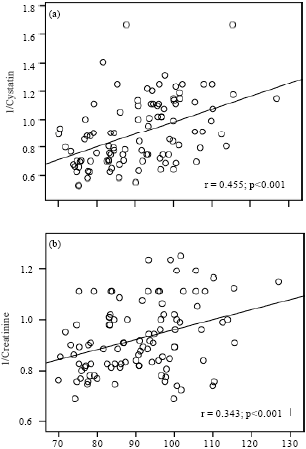
Research Article
Assessing Validity of Serum Cystatin C for Predicting Metabolic Syndrome
Department of Laboratory Medicine, Faculty of Paramedical Sciences, Maragheh, Tabriz, Iran
MirMoosa Mirinejad
Faculty of Medicine, Tabriz, Iran
Hooshang Amirrasooli
Mohammad Tagikhani
Faculty of Medicine, Tabriz, Iran