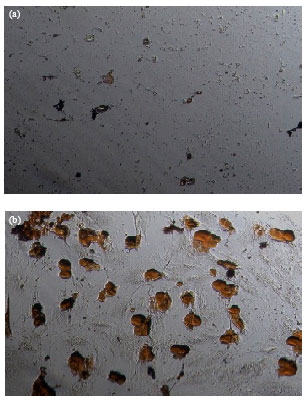
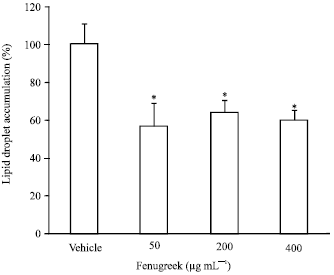
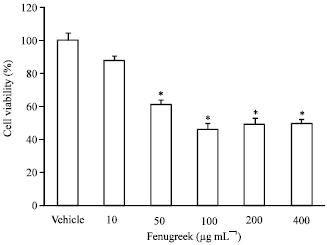
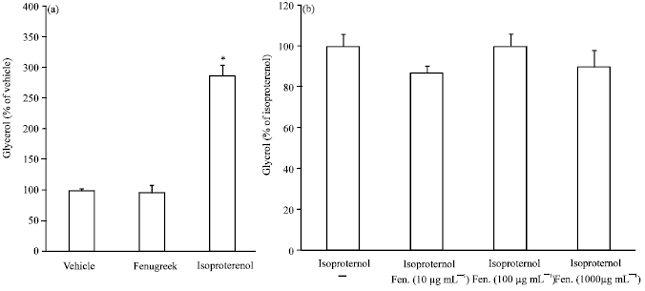
Research Article
Effects of Fenugreek Seeds on Adipogenesis and Lipolysis in Normal and Diabetic Rats
Pharmacological Research Center of Medicinal Plants, School of Medicine, Mashhad University of Medical Sciences, Mashhad, Iran
Mousa-Al-Reza Hadjzadeh
Neurocognitive Research Center, Department of Physiology, School of Medicine, Mashhad University of Medical Sciences, Mashhad, Iran
Ziba Rajaei
Department of Physiology, School of Medicine, Isfahan University of Medical Sciences, Isfahan, Iran
Seyed Bamdad Zendehbad
Neurocognitive Research Center, Department of Physiology, School of Medicine, Mashhad University of Medical Sciences, Mashhad, Iran