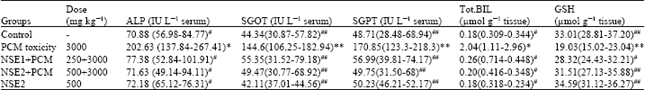
Research Article
Protective Effects of Ethanolic Extract of Nigella sativa Seed in Paracetamol Induced Acute Hepatotoxicity In vivo
Department of Pharmacology, Era`s Lucknow Medical College, Sarfarazganj, Hardoi Road, Lucknow, 226003, India
M.T. Salman
Department of Pharmacology, Era`s Lucknow Medical College, Sarfarazganj, Hardoi Road, Lucknow, 226003, India
P. Singh
Department of Pathology, Christian Medical College, Vellore, India
V.K. Verma
Department of Pharmacology, Era`s Lucknow Medical College, Sarfarazganj, Hardoi Road, Lucknow, 226003, India
A. Ahmad
Department of Pharmacology, Era`s Lucknow Medical College, Sarfarazganj, Hardoi Road, Lucknow, 226003, India