Research Article
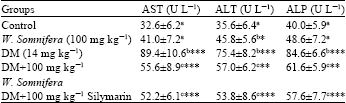
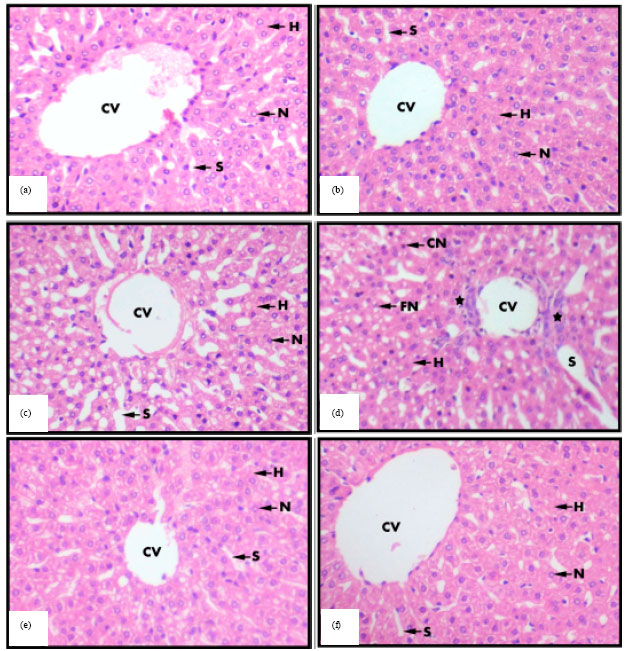
Effects of Aqueous Extract of Withania somnifera on Some Liver Biochemical and Histopathological Parameters in Male Guinea Pigs
Department of Biology, Faculty of Science, Ibb University, Ibb, 70270, Yemen
Samira M. Hezabr
Department of Biology, Faculty of Science, Ibb University, Ibb, 70270, Yemen
Aisha M. Al-Zubairi
Department of Biology, Faculty of Science, Ibb University, Ibb, 70270, Yemen
Faten A. Al-Hemiri
Department of Biology, Faculty of Science, Ibb University, Ibb, 70270, Yemen