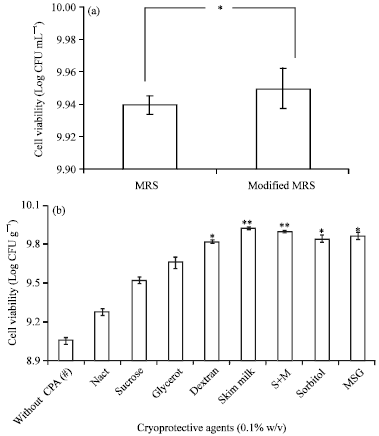
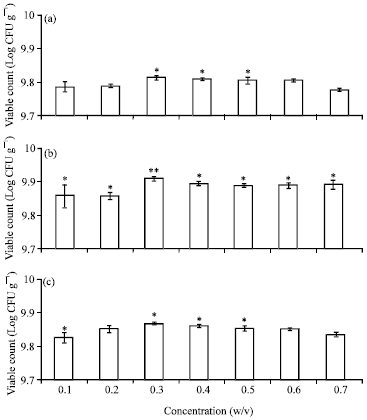
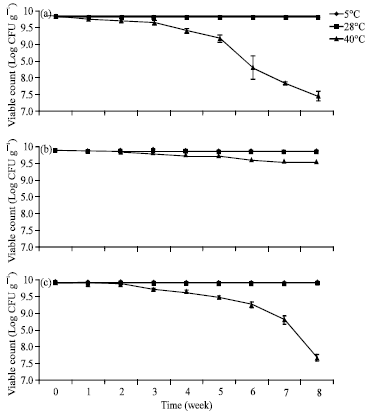
Research Article
Cost Effectiveness of Cryoprotective Agents and Modified De-man Rogosa Sharpe Medium on Growth of Lactobacillus acidophilus
Department of Pharmaceutical Technology, School of Pharmaceutical Sciences, Universiti Sains Malaysia, 11800 Minden, Penang, Malaysia
Kok-Khiang Peh
Department of Pharmaceutical Technology, School of Pharmaceutical Sciences, Universiti Sains Malaysia, 11800 Minden, Penang, Malaysia