Research Article
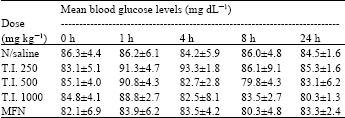
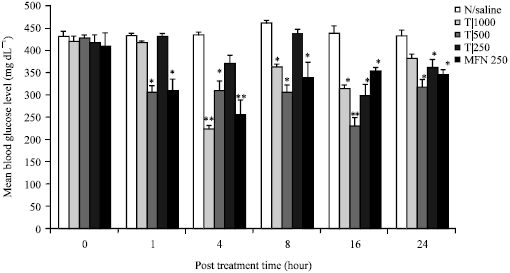
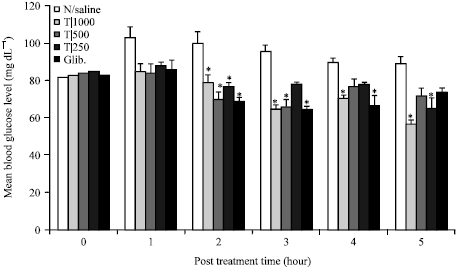
Antihyperglycaemic Activity of the Stem-Bark Extract of Tamarindus indica L. on Experimentally Induced Hyperglycaemic and Normoglycaemic Wistar Rats
Department of Pharmacology and Therapeutics Ahmadu Bello University, Zaria, Nigeria
J.A. Anuka
Department of Pharmacology and Therapeutics Ahmadu Bello University, Zaria, Nigeria
O.A. Salawu
Department of Pharmacology and Toxicology, National Institute for Pharmaceutical Research and Development Abuja, Nigeria
I. Abdu-Aguye
Department of Pharmacology and Therapeutics Ahmadu Bello University, Zaria, Nigeria