Research Article
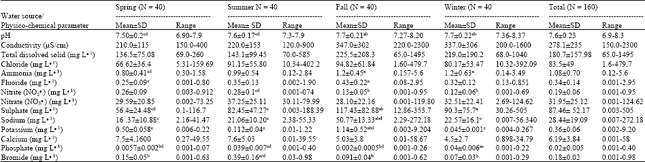
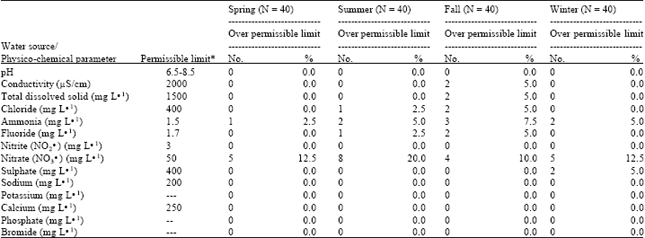
Physico-chemical Analysis of Drinking Groundwater of Around Tehran by Seasonal Variation
Departement of Biochemistry, Pasteur Institute of Iran, Tehran, Iran
S. Javadian
Departement of Biochemistry, Pasteur Institute of Iran, Tehran, Iran
Z. Zamani
Departement of Biochemistry, Pasteur Institute of Iran, Tehran, Iran
S.D. Siadat
Department of Microbiology, Pasteur Institute of Iran, Tehran, Iran
Sh- Khatami
Departement of Biochemistry, Pasteur Institute of Iran, Tehran, Iran
R. Saghiri
Departement of Biochemistry, Pasteur Institute of Iran, Tehran, Iran
T. Kordi
University of Tehran, Tehran, Iran