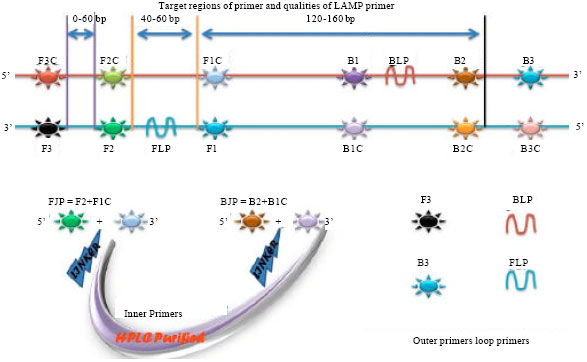
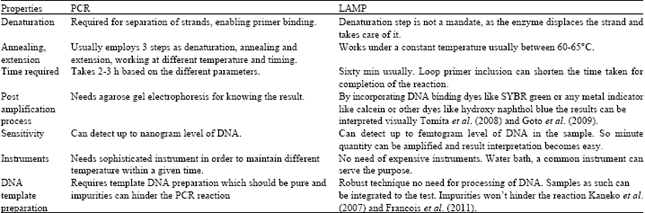
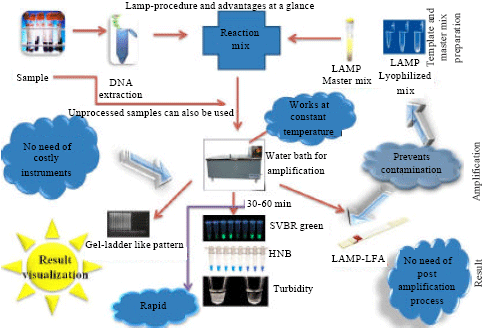
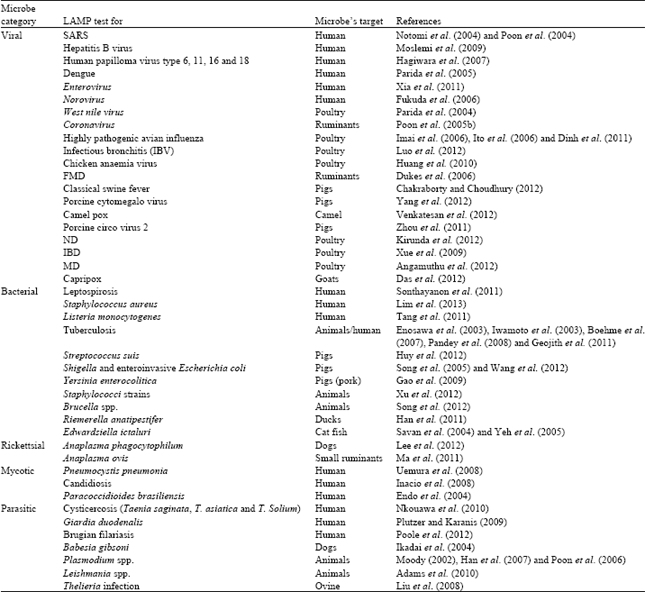
Review Article
Loop-mediated Isothermal Amplification of DNA (LAMP): A New Diagnostic Tool Lights the World of Diagnosis of Animal and Human Pathogens: A Review
Division of Pathology, Izatnagar, Bareilly (UP)-243122, India
K. Karthik
Division of Bacteriology and Mycology, Indian Veterinary Research Institute, Izatnagar, Bareilly (UP)-243122, India
Sandip Chakraborty
Animal Resources Development Department, Pt. Nehru Complex, Agartala, Tripura-799006, India
Ruchi Tiwari
Department of Veterinary Microbiology and Immunology, Uttar Pradesh Pandit Deen Dayal Upadhayay Pashu Chikitsa Vigyan Vishwa Vidyalaya Evum Go-Anusandhan Sansthan (DUVASU), Mathura (UP)-281001, India
Sanjay Kapoor
Department of Veterinary Microbiology, Lala Lajpat Rai University of Veterinary and Animal Sciences, Hisar, Haryana, India
Amit Kumar
Department of Veterinary Microbiology and Immunology, Uttar Pradesh Pandit Deen Dayal Upadhayay Pashu Chikitsa Vigyan Vishwa Vidyalaya Evum Go-Anusandhan Sansthan (DUVASU), Mathura (UP)-281001, India
Prasad Thomas
Division of Bacteriology and Mycology, Indian Veterinary Research Institute, Izatnagar, Bareilly (UP)-243122, India